DELMAR PHARMACEUTICALS, INC.
SUPPLEMENTAL COMPANY INFORMATION DATED JUNE 26, 2014
DelMar Pharmaceuticals, Inc. (the “Company”) is a Nevada corporation formed on June 24, 2009 under the name Berry Only Inc. (“Berry”). Prior to the Reverse Acquisition (discussed below), Berry did not have any significant assets or operations. On January 21, 2013, the Company changed its name to DelMar Pharmaceuticals, Inc.
DelMar Pharmaceuticals, Inc. is the parent company of Del Mar Pharmaceuticals (BC) Ltd. (“DelMar (BC)”), a British Columbia, Canada corporation incorporated on April 6, 2010, which is a clinical and commercial stage drug development company with a focus on the treatment of cancer. We are conducting clinical trials in the United States with our lead product, VAL-083, as a potential new treatment for GBM, the most common and aggressive form of brain cancer. We have also acquired certain exclusive commercial rights to VAL-083 in China where it is approved as a chemotherapy for the treatment of chronic myelogenous leukemia (“CML”) and lung cancer. We plan to seek marketing partnerships in China in order to generate royalty revenue.
Our executive offices are located at Suite 720-999 West Broadway, Vancouver, British Columbia, Canada V5Z 1K5. Our clinical operations are managed at Suite R, 3475 Edison Way, Menlo Park, California, 94025. Our website is located at www.delmarpharma.com, and our telephone number is 604-629-5989.
On January 25, 2013 (the “Closing Date”), the Company entered into and closed an exchange agreement (the “Exchange Agreement”), with DelMar (BC), 0959454 B.C. Ltd., a British Columbia corporation and a wholly-owned subsidiary of the Company (“Callco”), 0959456 B.C. Ltd., a British Columbia corporation and a wholly-owned subsidiary of the Company (“Exchangeco”), and securityholders of DelMar (BC). Pursuant to the Exchange Agreement, (i) the Company issued 4,340,417 shares of common stock (the “Parent Shares”) to the shareholders of DelMar (BC) who are United States residents (the “U.S. Holders”) in exchange for the transfer to Exchangeco of all 4,340,417 outstanding common shares of DelMar (BC) held by the U.S. Holders, (ii) the shareholders of DelMar (BC) who are Canadian residents (the “Canadian Holders”) received, in exchange for the transfer to Exchangeco of all 8,729,583 outstanding common shares of DelMar (BC) held by the Canadian Holders, 8,729,583 exchangeable shares (the “Exchangeable Shares”) of Exchangeco, and (iii) outstanding warrants to purchase 3,360,000 common shares of DelMar (BC) and outstanding options to purchase 1,020,000 common shares of DelMar (BC) were deemed to be amended such that, rather than entitling the holder to acquire common shares of DelMar (BC), such options and warrants (as amended, the “Exchange Agreement Warrants”) will entitle the holders to acquire shares of common stock of the Company. The Canadian Holders will be entitled to require Exchangeco to redeem (or, at the option of the Company or Callco, to have the Company or Callco purchase) the Exchangeable Shares, and upon such redemption or purchase to receive an equal number of shares of common stock of the Company.
Effective on the Closing Date, pursuant to the Exchange Agreement, DelMar (BC) became (indirectly through Exchangeco) a wholly-owned subsidiary of the Company. The acquisition of DelMar (BC) is treated as a reverse acquisition, and the business of DelMar (BC) became the business of the Company. At the time of the Reverse Acquisition, Berry was not engaged in any active business.
Our mission is to benefit patients and create shareholder value by rapidly developing and commercializing anti-cancer therapies in orphan cancer indications where patients have failed or are unlikely to respond to modern therapy. Our lead product candidate, VAL-083, represents a “first-in-class” small-molecule chemotherapeutic, which means that the molecular structure of VAL-083 is not an analogue or derivative of other small molecule chemotherapeutics approved for the treatment of cancer. VAL-083 has been assessed in multiple clinical studies sponsored by the National Cancer Institute (“NCI”) in the United States as a treatment against various cancers including lung, brain, cervical, ovarian tumors and leukemia. Published pre-clinical and clinical data suggest that VAL-083 may be active against a range of tumor types. VAL-083 is approved as a cancer chemotherapeutic in China for the treatment of chronic mylogenous leukemia (”CML”) and lung cancer. VAL-083 has not been approved for any indication outside of China.
Upon obtaining regulatory approval, we intend to commercialize VAL-083 for the treatment of orphan and other cancer indications where patients have failed other therapies or have limited medical options. Orphan diseases are defined in the United States under the Rare Disease Act of 2002 as “any disease or condition that affects less than 200,000 persons in the United States”. The Orphan Drug Act of 1983 is a federal law that provides financial and other incentives including a period of market exclusivity to encourage the development of new treatments for orphan diseases. In February 2012, we announced that VAL-083 has been granted protection under the Orphan Drug Act by the United States Food and Drug Administration (“FDA”) for the treatment of glioma, including GBM. In January 2013, the European Medicines Agency (“EMA”) also granted orphan drug protection to VAL-083 for the treatment of glioma.
We research the mechanism of action of our product candidate to determine the clinical indications best suited for therapy and work rapidly advance it into human clinical trials and toward commercialization. With this aim, in October 2011 we initiated clinical trials with VAL-083 as a potential new treatment for GBM, the most common and aggressive form of brain cancer. We have presented interim data from our clinical trial at peer reviewed scientific meetings demonstrating that VAL-083 can shrink or halt the growth of tumors in some brain cancer patients who have failed other approved treatments. Currently, there is no approved therapy for these patients.
In addition to our clinical development activities in the United States, we have obtained exclusive commercial rights to VAL-083 in China. In October 2012, we announced that we had entered into a collaboration agreement with the only manufacturer presently licensed by the China Food and Drug Administration (“CFDA”) to produce the product for the China market. This agreement provides us with exclusive commercial rights which potentially position us to generate near-term revenue through product sales or royalties for its approved indications in China while we seek global approval in new indications. We anticipate that we may be able to begin generating revenue from such sales or royalties commencing in 2014.
VAL-083 was originally discovered in the 1960’s. We have filed a broad portfolio of new patent applications to protect our intellectual property. Our patent applications claim compositions and methods related to the use of VAL-083 and related compounds as well as methods of synthesis and quality controls for the manufacturing process of VAL-083. In July 2013, our first patent was granted by the United States Patent and Trademark Office. The patent expiration date is August 17, 2031. In addition, VAL-083 has been granted protection under the Orphan Drug Act by the FDA and the EMA. We believe that our portfolio of intellectual property rights provides a strong and defensible market position for the commercialization of VAL-083 and other anti-cancer products.
We also believe the experience of our clinical development team will position us to acquire or license additional product candidates to establish a pipeline of product opportunities. We have secured three grants from the National Research Council of Canada, which have provided financial contributions of over Cdn $130,000 to date. We believe we have the potential to create significant value by building and maintaining a sustainable business through the commercialization of VAL-083 across a variety of cancer indications on a world-wide basis.
Our drug discovery research focuses on identifying well-validated clinical and commercial-stage compounds and establishing a scientific rationale for development in modern orphan drug indications. Through our relationship with Valent Technologies, LLC (“Valent”), a company owned by Dr. Dennis Brown, our Chief Scientific Officer, we are able to utilize Valent’s proprietary ChemState™ bioinformatics tools which are used to screen and identify potential candidates. Promising candidates are further researched through our network of consultants and contract research organizations. This approach allows us to rapidly identify and advance potential drug candidates without significant investment in “wet lab” infrastructure. Based on this strategy, we acquired initial VAL-083 intellectual property and prototype drug product from Valent and have identified multiple additional drug candidates that we may have the opportunity to license or acquire in the future.
VAL-083 is a novel “first in class” small-molecule therapeutic agent that we are developing as a new cancer chemotherapy.
VAL-083 has been assessed in multiple NCI sponsored clinical studies in various cancers including lung, brain, cervical, ovarian tumors and leukemia. Published pre-clinical and clinical data from the late 1970s and 1980s suggest that VAL-083 may be active against a range of tumor types; however, further research was not pursued in the United States due to an increased focus by the NCI on targeted biologic therapies during the era. VAL-083 is approved as a cancer chemotherapeutic in China for the treatment of CML and lung cancer.
The mechanism of action of VAL-083 is understood to be a bi-functional alkylating agent. Alkylating agents are a commonly used class of chemotherapy drugs. They work by binding to DNA and interfering with normal processes within the cancer cell, which prevents the cell from making the proteins needed to grow and survive. After exposure to alkylating agents, the cancer cell becomes dysfunctional and dies. There are a number of alkylating agents on the market that are used by physicians to treat different types of cancer.
Based on published research, the functional groups associated with the mechanism of action of VAL-083 are understood to be functionally different from commonly used alkylating agents, including Temodar®, which is commonly used a front-line chemotherapy against GBM, the most common and aggressive form of brain cancer. VAL-083 has previously demonstrated activity in cell-lines that are resistant to other types of chemotherapy. No evidence of cross-resistance has been reported in published clinical studies. Based on the presumed alkylating functionality of VAL-083, published literature suggests that DNA repair mechanisms associated with the leading brain cancer therapies, including Temodar ® and nitrosourea resistance, may not confer resistance to VAL-083. Therefore, we believe that VAL-083 may be effective in treating tumors that have failed or become resistant to other chemotherapies.
We have presented new research at peer-reviewed scientific meetings demonstrating that VAL-083 is active in some patients, patient-derived tumor cell lines and cancer stem cells that are resistant to other chemotherapies. Of particular importance is resistance to Temodar ® due to activity of the repair enzyme known as MGMT, which results in resistance to front-line therapy in many GBM patients. At AACR in 2012, we presented data demonstrating that VAL-083 is active independent of MGMT resistance in laboratory studies.
VAL-083 readily crosses the blood brain barrier where it maintains a long half-life in comparison to the plasma. Published preclinical and clinical research demonstrates that VAL-083 is selective for brain tumor tissue.
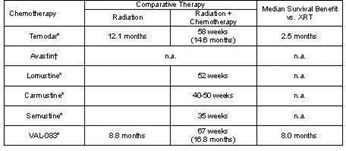
VAL-083 has been assessed in multiple studies as chemotherapy in the treatment of newly diagnosed and recurrent brain tumors and other cancers. In general, tumor regression in brain cancer was achieved following therapy in greater than 40% of patients treated and stabilization was achieved in an additional 20% - 30%. In published clinical studies, VAL-083 has previously been shown to have a statistically significant impact on median survival in high grade glioma brain tumors when combined with radiation vs. radiation alone.
A summary of published data adapted from separate sources comparing the efficacy of VAL-083 and other therapies in the treatment of glioblastoma multiforme (GBM).
The main dose-limiting toxicity (“DLT”) related to the administration of VAL-083 in previous NCI-sponsored clinical studies was myelosuppression. Myelosuppression is the decrease in cells responsible for providing immunity, carrying oxygen, and those responsible for normal blood clotting. Myelosuppression is a common side effect of chemotherapy. There is no evidence of lung, liver or kidney toxicity even with prolonged treatment by VAL-083. Commercial data from the Chinese market where the drug has been approved for more than 15 years supports the safety findings of the NCI studies.
We note that the DLT of VAL-083 was established prior to the development of medicines now available to manage myelosuppression. Various types of medications and other forms of therapy are now available for management of myelosuppressive side effects. We believe this offers the potential of increasing the dose of VAL-083 in the modern patient population thereby providing a potential opportunity to improve the drugs already established efficacy profile.
VAL-083 Clinical Development in GBM
Based on historical data and our own research, we filed an investigational new drug (“IND”) application with the FDA and initiated human clinical trials with VAL-083 as a potential treatment for GBM in 2011.
Our clinical trial is a Phase I/II an open-label, single arm dose-escalation study designed to evaluate the safety, tolerability, pharmacokinetics and anti-cancer activity of VAL-083 in patients with GBM. To be eligible for our clinical trial, patients must have been previously treated for GBM with surgery and/or radiation, if appropriate, and must have failed both Bevacizumab (Avastin ®) and temozolomide (Temodar ®), unless either or both are contra-indicated. .
Response to treatment with VAL-083 is measured prior to each treatment cycle. An initial phase of the study involves dose escalation cohorts until a maximum tolerated dose (“MTD”) is established in the context of modern care. The goal of our Phase I/II clinical trial is to determine a modernized dosing regimen for advancement into a registration directed clinical trial.
In February 2012, we announced that VAL-083 was granted protection under the Orphan Drug Act by the FDA for the treatment of glioma. In January 2013, we announced that the European Union had also granted orphan drug protection to VAL-083. Orphan drugs generally follow the same regulatory development path as any other pharmaceutical product. However, incentives such as scientific advice and reduction or waiver of registration fees and access to specialized grant funding may be available to support and accelerate development of orphan drug candidates. In addition, DelMar Pharma may sell VAL-083 as a treatment for glioma without competition for seven years in the US and for ten years in the EU following market approval, in respect of a medicinal product containing a similar active substance for the same indication.
Based on historical development of other products in GBM, we believe that we may be able to obtain FDA approval to commercialize VAL-083 to treat patients who have failed other therapies from an open-label Phase II registration-directed clinical, which will save significant costs of a large Phase III clinical trial. We also believe that the FDA may grant fast-track, accelerated approval and/or priority review status to VAL-083, which will enable us to begin filing for commercial approval during the clinical trial process. Fast Track, Accelerated Approval and Priority Review are approaches established by the FDA that are intended to make therapeutically important drugs available at an earlier time. (See “Government Regulation and Product Approval”.)
We are conducting the study under the direction of Dr. Howard Burris at the Sarah Cannon Research Institute in Nashville, Tennessee with a second center in Sarasota, Florida. In July 2013, the Company announced the opening of its third clinical trial site at the Brain Tumor Center at University of California, San Francisco (“UCSF”).
We have presented interim data from our clinical trial at peer-reviewed scientific meetings including the Society for NeuroOncology annual meeting (“SNO” – November, 2012), the American Association of Cancer Research (“AACR” – April 2013), the American Society for Clinical Oncology (“ASCO” – June 2013), the World Federation of Neuro‐Oncology (“WFNO” – November, 2013), AACR in April 2014 and ASCO in May 2014. In summary, our interim clinical data supports that:
| ● |
|
To date, one of two GBM patients in cohort 6 (30 mg/m2) exhibited stable disease after one cycle of treatment. Outcomes and analysis of cohorts 6 and 7 are ongoing; |
| |
|
|
| ● |
|
In earlier cohorts, DelMar reported that two patients exhibited a response (stable disease or partial response) with a maximum response of 28 cycles (84 weeks) and improved clinical signs prior to discontinuing due to adverse events unrelated to study; |
| |
|
|
| ● |
|
No drug-related serious adverse events have been detected, and maximum tolerated dose (“MTD”) has not been reached at doses up to 30 mg/m2. Enrollment and evaluation of Cohort 7 (40mg/m2) is ongoing; |
| |
|
|
| ● |
|
DelMar has also presented data demonstrating that the cytotoxic activity of VAL-083 is independent of MGMT, the enzyme believed to cause resistance to the current front-line therapy in the treatment of GBM; and |
| |
|
|
| ● |
|
Pharmacokinetics are linear and consistent with previous published data suggesting that concentrations of VAL-083 being obtained are effective against glioma cell lines in vitro. |
These data support the further development of VAL-083. We are continuing with the dose escalation portion of our clinical trial and anticipate achieving the maximum tolerated dose during 2014.
In August 2013 the Company received a notice of allowance from the FDA enabling the Company to implement a more rapid dose-escalation scheme in our GBM study. The revised dosing regimen was allowed by the FDA following an extensive safety review of patients treated to date. In comparison to the original dose-escalation scheme, the revised plan will enable the trial to reach higher doses and complete the dose-escalation portion of the clinical trial more quickly by skipping two interim doses.
A summary of our original and revised dose escalation scheme including doses completed to date is as follows:
|
Dose Escalation Scheme (mg/m2)
|
|
|
|
|
|
|
Original
|
|
|
Revised
|
|
|
Patients Treated
|
|
Status
|
| |
1.5 |
|
|
|
1.5 |
|
|
|
3 |
|
Completed – No Dose Limiting Toxicity, or “DLT”
|
| |
3.0 |
|
|
|
3.0 |
|
|
|
4 |
* |
Completed – No DLT
|
| |
5.0 |
|
|
|
5.0 |
|
|
|
10 |
* |
Completed – No DLT
|
| |
10.0 |
|
|
|
10.0 |
|
|
|
3 |
|
Completed – No DLT
|
| |
15.0 |
|
|
|
|
|
|
|
|
|
|
| |
20.0 |
|
|
|
20.0 |
|
|
|
3 |
|
Completed – No DLT
|
| |
25.0 |
|
|
|
|
|
|
|
|
|
|
| |
30.0 |
|
|
|
30.0 |
|
|
|
3 |
|
Completed – No DLT
|
|
n.a
|
|
|
|
40.0 |
|
|
3(planned)
|
|
Initiated May 2014
|
|
*Cohorts 2 and 3 were expanded to allow for patient demand and to gather additional data on CNS metastases patients.
|
If the MTD is not reached in cohort 7, DelMar would be prepared to file a protocol amendment with the FDA to allow dosing beyond 40mg/m2. During the remainder of 2014 we plan to continue our clinical trials with VAL-083 as a potential treatment for GBM patients who have failed other therapies. Currently, there is no approved therapy for these patients. The goal of the current trial is to establish a modernized dosing regimen for advancement into registration directed trials in the United States as a potential new therapy for the treatment of refractory GBM.
As part of our ASCO presentation on June 1, 2013, we also announced that we plan to split our current clinical trial protocol into two separate studies: one focusing solely on refractory GBM and the other focusing on secondary brain cancers caused by other tumors that have spread to the brain. Due to prior chemotherapy and radiation therapy, patients with secondary brain tumors are likely more prone to myelosuppression and may have a different toxicity and MTD than patients with GBM. We believe the strategy of splitting the trial into two separate studies will enable us to focus on accelerating the development of VAL-083 as a potential new treatment for glioblastoma while appropriately exploring the potential of the drug to treat patients with solid tumors that have spread to the brain.
We anticipate presenting additional data at upcoming scientific meetings during 2014.
The current study is being conducted under an IND application with the FDA. It involves a dose-escalation phase (Phase I) and an efficacy phase (Phase II). Phase I of the study will continue to enroll patients until a MTD is achieved. Based on historical data, we anticipate that Phase I will involve up to 30 patients. An additional 14 GBM patients can be enrolled at the MTD or a lower dose recommended by the principal investigator. Details of the study, including enrollment estimates, are available at http://www.clinicaltrials.gov/ct2/show/NCT01478178?term=VAL-083&rank=1). We plan to develop a separate protocol for the continued exploration of VAL-083 in patients with secondary brain cancer caused by solid tumor spreading to the brain.
While our data with VAL-083 to date are interim in nature, we believe the results to date demonstrate a strong potential for successful development of VAL-083 as a chemotherapy for the treatment of GBM. We plan to continuing working with our clinical investigators to determining an optimal dosing regimen for future registration trials.
VAL-083 in Leukemia and Hematologic Cancers
CML, also known as chronic myeloid leukemia, is a cancer of the white blood cells. The incidence of CML in the United States is approximately two per 100,000 population.
CML is characterized by three progressive phases: chronic, aggressive and blast, each corresponding with poorer prognosis. Approximately 85% of patients with CML are in the chronic phase at the time of diagnosis. Chronic phase patients are usually asymptomatic or have only mild symptoms such as fatigue or no symptoms at all. The duration of chronic phase is variable and depends on how early the disease was diagnosed as well as type of treatment. Without treatment, CML progresses to an accelerated phase and eventually to blast crisis. Blast crisis is the final phase in the evolution of CML and behaves like an acute leukemia with rapid progression and short expected survival.
VAL-083 has shown promise in CML in multiple pre-clinical and clinical studies. The NCI studied VAL-083 extensively in laboratory and animal models of hematological malignancies (blood cancers). VAL-083 has been approved for the treatment of CML in China. While VAL-083 maintains labeling for CML in China, use of the drug in the modern era has been limited by a preference for targeted therapies such as tyrosine kinase inhibitors (TKIs).
TKIs have become the standard of care for CML and non-small cell lung cancer (NSCLC). TKI therapy has resulted in vastly improved outcomes; however, patients often develop resistance to TKI therapy. Recent evidence proposes unique mechanisms of resistance in patients of East Asian descent who experience significantly inferior responses to TKIs, including imatinib (Gleevec ®) in CML and erlotinib (Tarceva ®) in lung cancer.
We believe that data from NCI-sponsored studies and commercial evidence from the Chinese market support substantive clinical benefit of VAL-083 in CML. We also believe that the unique mechanism of action of VAL-083, in combination with newly developed data positions the drug as a valuable therapy for patients who have failed other treatments, including TKIs. This represents a significant clinical and commercial opportunity for large subsets of patient populations in the existing-approved China market as well as for global development in CML.
Based on these beliefs, we have acquired certain commercial rights to VAL-083 in China where it is approved for the treatment of CML and Lung Cancer. We have also developed new non-clinical data demonstrating that VAL-083 is active against TKI-resistant CML. We have begun to establish a network of leading oncologists to develop new clinical and non-clinical data which will demonstrate the clinical utility of VAL-083 in CML patients who are resistant to TKIs. We believe this strategy will result in sales growth for VAL-083 in China and generate near-term revenue for our company through sales and marketing partnerships as well as position VAL-083 for global development in CML.
In addition, we plan to investigate VAL-083 as a potential treatment for other types of blood cancer. Acute Myeloid Leukemia (“AML”) and Acute Lymphoblastic Leukemia (“ALL”) are of particular interest based on published data and lack of effective therapeutic options. We have initiated preliminary discussions with leading cancer centers regarding the development of a clinical strategy for the development of VAL-083 in other types of blood cancer.
Lung cancer is characterized as small cell and non-small cell lung cancer (“NSLSC”). NSCLC is the most common type of lung cancer.
There are three common forms of NSCLC: adenocarcinomas are often found in an outer area of the lung; squamous cell carcinomas are usually found in the center of the lung next to an air tube (bronchus); and large cell carcinomas, which can occur in any part of the lung and tend to grow and spread faster than adenocarcinoma.
Smoking is the most important risk factor in the development of lung cancer. According to the World Cancer Report (2008), 21% of cancer deaths are related to smoking, especially lung cancer. Additionally, high levels of air pollution have been implicated as significant causes of lung cancer. Incidence of lung cancer in the United States is approximately 59 per 100,000 with the majority (52:100,000) being NSLSC.
According to The Nationwide Nutrition and Health Survey (2002), China has the world’s largest smoking population, with a smoking rate of 24.0% on average (50.2% for men and 2.8% for women), and a total number of 350 million smokers. The World Health Organization reports that the incidence of lung cancer in China is 34 per 100,000 population. However, some estimates are much higher exceeding 120 per 100,000 population for males aged 55-60 in urban areas.
According to a survey conducted by the Chinese Ministry of Health and the Ministry of Science and Technology, smoking, poor diet, water pollution and environmental problems have caused the nation's cancer death rate to rise 80 percent in the past 30 years and cancer is now accountable for 25 percent of all urban deaths and 21 percent of all rural deaths. Based on these trends, the World Health Organization projects that the incidence of lung cancer in China is expected to exceed one million (1,000,000) new cases per year by 2025.
Similar to CML treatment, TKIs are standard front-line therapy in certain types of NSCLC; however resistance to TKI therapy is common in lung cancer patients. It has also been reported that cigarette smoke may directly induce resistance to TKIs. This factor could further exacerbate resistance to modern targeted therapies in populations such as China where smoking is highly prevalent. In addition, the same East-Asian specific resistance linked to TKI-resistance in CML has been shown to correlate with TKI-resistance in NSLSC.
The activity of VAL-083 against lung cancer was studied extensively by the NCI. VAL-083 demonstrated activity against NSCLC in laboratory and animal studies. VAL-083 was also investigated in a number of clinical trials in the United States and Europe during the 1970s both as a stand-alone therapy and in combination with other chemotherapeutic regimens. VAL-083 has been approved for the treatment of lung cancer in China; however, we believe that the use of the drug in the modern era has been limited by a preference for targeted therapies such as TKIs.
We believe VAL-083’s unique bi-functional alkylating mechanism of action could make it a valuable drug of choice in NSCLC patients who are or become resistant to TKI therapy. In addition, VAL-083 readily crosses the blood brain barrier suggesting that it may be possible for VAL-083 to treat patients whose lung cancer has spread to the brain.
Based on these beliefs, we have acquired certain commercial rights to VAL-083 in China where it is approved for the treatment of lung cancer. We plan to work with leading oncologists to develop new clinical and non-clinical data which will demonstrate the clinical utility of VAL-083 in NSCLC patients who are resistant to TKIs. We believe this strategy will result in sales growth for VAL-083 in China and generate near-term revenue for our company through sales and marketing partnerships as well as position VAL-083 for global development in lung cancer.
In April 2014 at AACR we announced results of pre-clinical study designed to evaluate the activity of VAL-083 in in vivo models of drug-resistant NSCLC in comparison to cisplatin.
In an established murine xenograft model of NSCLC, the activity of VAL-083 was compared to standard platinum-based therapy with cisplatin against human NSCLC cell lines A549 (TKI-sensitive) and H1975 (TKI-resistant). In the study, VAL-083 demonstrated superior efficacy and safety in the treatment of TKI-susceptible (A549) tumors and in TKI-resistant (H1975) tumors.
|
·
|
Treatment of TKI-sensitive (A549) NSCLC with 3 mg/kg of VAL-083 resulted in tumor growth delay of 26 days compared to untreated controls. Cisplatin (5 mg/kg) resulted in tumor growth delay of just four days. In addition, mean tumor volume on day 68 was significantly reduced in animals treated with 3 mg/kg VAL-083 (p=0.001) compared to untreated control.
|
|
·
|
Treatment of TKI-resistant (H1975) NSCLC with 4 mg/kg of VAL-083 resulted in a statistically significant reduction in tumor volume (p = 0.01) versus untreated control after 27 days. In the same model, treatment with 5 mg/kg of cisplatin failed to achieve statistically significant reduction in tumor volume (p = 0.23) versus untreated control after 27 days. Longer-term safety assessments are ongoing in this model.
|
These data suggest that VAL-083 may be a viable treatment option for NSCLC patients failing TKI-therapy, especially where platinum-based therapy has already failed or is predicted to give sub-optimal outcomes. These results may have immediate implications in the treatment of NSCLC in China, where VAL-083 is approved for as a chemotherapy for the treatment of lung cancer. The data also support exploring future clinical development of VAL-083 as a lung cancer therapy in the rest of the world thereby providing DelMar with a potential opportunity to expand our clinical development focus beyond glioblastoma.
VAL-083 Target Markets
We are targeting cancer indications which we believe represent market opportunities in the hundreds of millions of dollars in North America and potentially in the billions of dollars worldwide. The pharmaceutical industry, in general, is a highly profitable, highly innovative industry. In 2006, the global pharmaceutical industry generated over $640 billion dollars in revenue. According to published reports, global pharmaceutical sales are highly stratified by region, with North America, the European Union and Japan accounting for 55% of global pharmaceutical sales in 2009; however, the most rapid growth in the sector is from developing countries, particularly China.
|
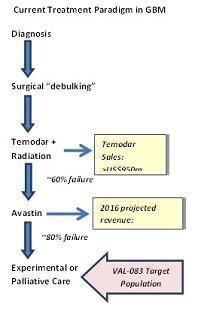
Glioblastoma Multiforme (GBM): Newly diagnosed patients suffering from GBM are initially treated through invasive brain surgery, although disease progression following surgical resection is nearly 100%. Temozolomide (Temodar ®) in combination with radiation is the front-line therapy for GBM following surgery. Temodar currently generates more than US$950 million annually in global revenues even though most patients fail to gain long-term therapeutic benefits. Approximately 60% of GBM patients treated with Temodar experience tumor progression within one year.
Bevacizumab (Avastin®) has been approved for the treatment of GBM in patients failing Temodar ®. In clinical studies, only about 20% of patients failing Temodar respond to Avastin therapy. In spite of these low efficacy results, treatment of GBM in North America alone is projected to add US$200 million annually to the revenues of Avastin with projected growth in GBM to US$650 million by 2016.
Approximately 48% of patients who are diagnosed with GBM will fail both front-line therapy and Avastin. Based on disease incidence, we believe the market for treating GBM patients the post-Avastin failure exceeds US$200 million annually in North America. Subject to successfully completing clinical trials and obtaining approval by the FDA and other applicable regulatory agencies globally, we also believe that VAL-083 could potentially generate sales in excess of $1 billion world-wide as a potential front-line therapy for GBM.
|
|
|
Leukemia: The potential of VAL-083 in the treatment of CML has been established in both human clinical trials conducted by the NCI and by the drug’s commercial approval in China. The Tyrosine Kinase Inhibitor Gleevec® is currently used as front-line therapy in the treatment of CML currently achieves global revenue in excess of $1 billion annually. We believe that VAL-083 has potential to capture a portion of the CML market through demonstration of activity in TKI-resistant CML patients. We also believe that VAL-083 may offer significant commercial opportunities through the treatment of other types of blood cancer such as AML or ALL.
Lung Cancer: The potential of VAL-083 in the treatment of NSLSC has been established in both human clinical trials conducted by the NCI and by the drug’s commercial approval in China. A 2012 report published by Decision Resources, Inc. (http://decisionresources.com/), forecasts that the NSCLC drug market will exceed US$4 billion in 2015.
VAL-083 is currently manufactured in accordance with CFDA and Chinese Pharmacopoeia guidelines to ensure drug quality control, drug use safety, and drug efficacy. Approval by the FDA will require VAL-083 and other products developed by us to be manufactured in accordance with United States Pharmacopeia (“USP”) in accordance with Good Manufacturing Practices (“cGMP”) regulations. cGMP provides for systems that assure proper design, monitoring, and control of manufacturing processes and facilities. Adherence to the cGMP regulations assures the identity, strength, quality, and purity of drug products by requiring that manufacturers of medications adequately control manufacturing operations.
We have established an exclusive purchasing relationship with the Chinese manufacturer that has enabled us to obtain drug product for human clinical trials in the United States and certain commercial rights in China. The Chinese manufacturer has established a commercial-scale manufacturing process based on the North American process originally developed for the NCI.
Ensuring a viable long-term supply of the VAL-083 drug product suitable for registration and commercialization in North America and Europe will require investment in improved manufacturing and quality controls. We will seek to build upon our expertise and our intellectual property related to the existing manufacturing processes for VAL-083 in collaboration with the current manufacturer to allow compliance with cGMP. In addition, we have identified third party contract manufacturers with the capabilities to establish the processes, procedures and quality systems necessary to meet U.S., Canadian, E.U. and other international cGMP manufacturing requirements. Such requirements include strong quality management systems, obtaining appropriate quality raw materials, establishing robust operating procedures, detecting and investigating product quality deviations, and maintaining reliable testing laboratories.
Patents and Proprietary Rights
Our success will depend in part on our ability to protect our existing product candidates and the products we acquire or license by obtaining and maintaining a strong proprietary position. To develop and maintain our position, we intend to continue relying upon patent protection, orphan drug status, Hatch-Waxman exclusivity, trade secrets, know-how, continuing technological innovations and licensing opportunities. We intend to seek patent protection whenever available for any products or product candidates and related technology we acquire in the future.
We have filed patent applications covering VAL-083 where we have claimed the use of and improvements related VAL-083 and other novel aspects of our proposed treatment regimen. We have also developed and filed patents on manufacturing process improvements for VAL-083. In addition, we plan to implement strategies which may enable us to acquire patent protection for the formulation and composition of the active pharmaceutical ingredient and finished dosage form of VAL-083 products. In July 2013, our first patent was granted by the United States Patent and Trademark Office. We are prosecuting all of our patent applications in the United States and in international jurisdictions which we deem important for the potential commercial success of VAL-083.
We may also seek orphan drug status whenever it is available. If a product which has an orphan drug designation subsequently receives the first regulatory approval for the indication for which it has such designation, the product is entitled to orphan exclusivity, meaning that the applicable regulatory authority may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for a period of seven years in the U.S. and Canada, and 10 years in the E.U. Orphan drug designation does not prevent competitors from developing or marketing different drugs for the same indication or the same drug for a different clinical indication. In February 2012, we announced that the FDA has granted orphan drug status to VAL-083. In January 2013, the EMA also granted orphan drug protection to VAL-083 for the treatment of glioma.
Under the Hatch-Waxman Amendments, newly approved drugs and indications benefit from a statutory period of non-patent marketing exclusivity. These amendments provide five-year data exclusivity to the first applicant to gain approval of an NDA for a new chemical entity, meaning that the FDA has not previously approved any other new drug containing the same active ingredient. The Hatch-Waxman Amendments prohibit the submission of an abbreviated new drug application, also known as an ANDA or generic drug application, during the five-year exclusive period if no patent is listed. If there is a patent listed and the ANDA applicant certifies that the NDA holder’s listed patent for the product is invalid or will not be infringed, the ANDA can be submitted four years after NDA approval. Protection under the Hatch-Waxman Amendments will not prevent the filing or approval of another full NDA; however, the applicant would be required to conduct its own pre-clinical studies and adequate and well-controlled clinical trials to demonstrate safety and effectiveness. The Hatch-Waxman Amendments also provide three years of data exclusivity for the approval of NDAs with new clinical trials for previously approved drugs and supplemental NDAs, for example, for new indications, dosages or strengths of an existing drug, if new clinical investigations were conducted by or on behalf of the sponsor and were essential to the approval of the application. This three-year exclusivity covers only the new changes associated with the supplemental NDA and does not prohibit the FDA from approving ANDAs for drugs containing the original active ingredient. We intend to rely on the Hatch-Waxman Amendments for five years of data exclusivity for VAL-083.
We also rely on trade secret protection for our confidential and proprietary information. We believe that the substantial costs and resources required to develop technological innovations, such as the manufacturing processes associated with VAL-083, will help us to protect the competitive advantage of our product candidates.
The protection of intellectual property rights in China (where our lead product candidate, VAL-083, is manufactured pursuant to a collaboration agreement with the only manufacturer presently licensed by the CFDA to produce the product for the China market, and where VAL-03 is approved for the treatment of CML and lung cancer) is relatively weak compared to the United States, which may negatively affect our ability to generate revenue from VAL-083.
It is our policy to require our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us. These agreements provide that all confidential information developed or made known to the individual during the course of the individual’s relationship with us is to be kept confidential and not disclosed to third parties except in specific circumstances. In the case of employees, the agreements provide that all inventions conceived by the individual shall be our exclusive property.
Government Regulation and Product Approval
Regulation by governmental authorities in the U.S. and other countries is a significant factor, affecting the cost and time of our research and product development activities, and will be a significant factor in the manufacture and marketing of any approved products. All of our products require regulatory approval by governmental agencies prior to commercialization. In particular, our products are subject to rigorous pre-clinical and clinical testing and other approval requirements by the FDA and similar regulatory authorities in other countries. Various statutes and regulations also govern or influence the manufacturing, safety, reporting, labeling, transport and storage, record keeping and marketing of our products. The lengthy process of seeking these approvals, and the subsequent compliance with applicable statutes and regulations, require the expenditure of substantial resources. Any failure by us to obtain, or any delay in obtaining, the necessary regulatory approvals could harm our business.
The regulatory requirements relating to the testing, manufacturing and marketing of our products may change from time to time and this may impact our ability to conduct clinical trials and the ability of independent investigators to conduct their own research with support from us.
The clinical development, manufacturing and marketing of our products are subject to regulation by various authorities in the U.S., the E.U. and other countries, including, in the U.S., the FDA, in Canada, Health Canada, and, in the E.U., the EMA. The Federal Food, Drug, and Cosmetic Act, the Public Health Service Act in the U.S. and numerous directives, regulations, local laws and guidelines in Canada and the E.U. govern the testing, manufacture, safety, efficacy, labeling, storage, record keeping, approval, advertising and promotion of our products. Product development and approval within these regulatory frameworks takes a number of years and involves the expenditure of substantial resources.
Regulatory approval will be required in all the major markets in which we seek to develop our products. At a minimum, approval requires the generation and evaluation of data relating to the quality, safety, and efficacy of an investigational product for its proposed use. The specific types of data required and the regulations relating to this data will differ depending on the territory, the drug involved, the proposed indication and the stage of development.
In general, new chemical entities are tested in animals until adequate evidence of safety is established to support the proposed clinical study protocol designs. Clinical trials for new products are typically conducted in three sequential phases that may overlap. In Phase I, the initial introduction of the pharmaceutical into either healthy human volunteers or patients with the disease (20 to 50 subjects), the emphasis is on testing for safety (adverse effects), dosage tolerance, metabolism, distribution, excretion and clinical pharmacology. Phase II involves studies in a limited patient population (50 to 200 patients) to determine the initial efficacy of the pharmaceutical for specific targeted indications, to determine dosage tolerance and optimal dosage and to identify possible adverse side effects and safety risks. Once a compound shows preliminary evidence of some effectiveness and is found to have an acceptable safety profile in Phase II evaluations, Phase III trials are undertaken to more fully evaluate clinical outcomes in a larger patient population in adequate and well-controlled studies designed to yield statistically sufficient clinical data to demonstrate efficacy and safety.
In the U.S., specific pre-clinical data, manufacturing and chemical data, as described above, need to be submitted to the FDA as part of an IND application, which, unless the FDA objects, will become effective 30 days following receipt by the FDA. Phase I studies in human volunteers may commence only after the application becomes effective. Prior regulatory approval for human healthy volunteer studies is also required in member states of the E.U. Currently, in each member state of the E.U., following successful completion of Phase I studies, data are submitted in summarized format to the applicable regulatory authority in the member state in respect of applications for the conduct of later Phase II studies. The regulatory authorities in the E.U. typically have between one and three months in which to raise any objections to the proposed study, and they often have the right to extend this review period at their discretion. In the U.S., following completion of Phase I studies, further submissions to regulatory authorities are necessary in relation to Phase II and III studies to update the existing IND. Authorities may require additional data before allowing the studies to commence and could demand that the studies be discontinued at any time if there are significant safety issues. In addition to the regulatory review, a study involving human subjects has to be approved by an independent body. The exact composition and responsibilities of this body will differ from country to country. In the U.S., for example, each study will be conducted under the auspices of an independent institutional review board at each institution at which the study is conducted. This board considers among other things, the design of the study, ethical factors, the privacy of protected health information as defined under the Health Insurance Portability and Accountability Act, the safety of the human subjects and the possible liability risk for the institution. Equivalent rules to protect subjects’ rights and welfare apply in each member state of the E.U. where one or more independent ethics committees, which typically operate similarly to an institutional review board, will review the ethics of conducting the proposed research. Other regulatory authorities around the rest of the world have slightly differing requirements involving both the execution of clinical trials and the import/export of pharmaceutical products. It is our responsibility to ensure we conduct our business in accordance with the regulations of each relevant territory.
By leveraging existing pre-clinical and clinical data, we are seeking build upon an existing pre-clinical and clinical safety and efficacy database to accelerate our research. In addition, our focus on end-stage population which has no current treatment options, commercialization may be achieved in an accelerated manner. Approval by the FDA in this category generally has been based on objective response rates and duration of responses rather than demonstration of survival benefit. As a result, trials of drugs to treat end-stage refractory cancer indications have historically involved fewer patients and generally have been faster to complete than trials of drugs for other indications. We are aware that the FDA and other similar agencies are regularly reviewing the use of objective endpoints for commercial approval and that policy changes may impact the size of trials required for approval, timelines and expenditures significantly.
In order to gain marketing approval we must submit a dossier to the relevant authority for review, which is known in the U.S. as an NDA and in the E.U. as a marketing authorization application, or MAA. The format is usually specific and laid out by each authority, although in general it will include information on the quality of the chemistry, manufacturing and pharmaceutical aspects of the product as well as the non-clinical and clinical data. Once the submitted NDA is accepted for filing by the FDA, it undertakes the review process that takes 10 months, unless an expedited priority review is granted which takes six months to complete. Approval can take several months to several years, if multiple 10-month review cycles are needed before final approval is obtained, if at all.
The approval process can be affected by a number of factors. The NDA may be approvable requiring additional pre-clinical, manufacturing data or clinical trials which may be requested at the end of the 10 month NDA review cycle, thereby delaying marketing approval until the additional data are submitted and may involve substantial unbudgeted costs. The regulatory authorities usually will conduct an inspection of relevant manufacturing facilities, and review manufacturing procedures, operating systems and personnel qualifications. In addition to obtaining approval for each product, in many cases each drug manufacturing facility must be approved. Further inspections may occur over the life of the product. An inspection of the clinical investigation sites by a competent authority may be required as part of the regulatory approval procedure. As a condition of marketing approval, the regulatory agency may require post-marketing surveillance to monitor for adverse effects or other additional studies as deemed appropriate. After approval for the initial indication, further clinical studies are usually necessary to gain approval for any additional indications. The terms of any approval, including labeling content, may be more restrictive than expected and could affect the marketability of a product.
The FDA offers a number of regulatory mechanisms that provide expedited or accelerated approval procedures for selected drugs in the indications on which we are focusing our efforts. These include accelerated approval under Subpart H of the agency’s NDA approval regulations, fast track drug development procedures and priority review. At this time, we have not determined whether any of these approval procedures will apply to our current drug candidate.
The U.S., E.U. and other jurisdictions may grant orphan drug designation to drugs intended to treat a “rare disease or condition,” which, in the U.S., is generally a disease or condition that affects no more than 200,000 individuals. In the E.U., orphan drug designation can be granted if: the disease is life threatening or chronically debilitating and affects no more than 50 in 100,000 persons in the E.U.; without incentive it is unlikely that the drug would generate sufficient return to justify the necessary investment; and no satisfactory method of treatment for the condition exists or, if it does, the new drug will provide a significant benefit to those affected by the condition. If a product that has an orphan drug designation subsequently receives the first regulatory approval for the indication for which it has such designation, the product is entitled to orphan exclusivity, meaning that the applicable regulatory authority may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for a period of seven years in the U.S. and 10 years in the E.U. Orphan drug designation does not prevent competitors from developing or marketing different drugs for the same indication or the same drug for different indications. Orphan drug designation must be requested before submitting an NDA or MAA. After orphan drug designation is granted, the identity of the therapeutic agent and its potential orphan use are publicly disclosed. Orphan drug designation does not convey an advantage in, or shorten the duration of, the review and approval process. However, this designation provides an exemption from marketing and authorization (NDA) fees.
We are also subject to numerous environmental and safety laws and regulations, including those governing the use and disposal of hazardous materials. The cost of compliance with and any violation of these regulations could have a material adverse effect on our business and results of operations. Although we believe that our safety procedures for handling and disposing of these materials comply with the standards prescribed by state and federal regulations, accidental contamination or injury from these materials may occur. Compliance with laws and regulations relating to the protection of the environment has not had a material effect on our capital expenditures or our competitive position. However, we are not able to predict the extent of government regulation, and the cost and effect thereof on our competitive position, which might result from any legislative or administrative action pertaining to environmental or safety matters.
The development and commercialization of new drugs is highly competitive and we may face competition established pharmaceutical and biotechnology companies, as well as from academic institutions, government agencies and private and public research institutions worldwide.
Various products currently are marketed for the treatment of GBM and other cancers that we may target with our product candidates and a number of companies are developing new treatments. Companies also developing products for GBM include but are not limited to Celgene Corp., Celldex Therapeutics, Northwest Biotherapeutics, Inc., Immunocellular Therapeutics Ltd., and many major pharmaceutical companies. Our success will be based in part on our ability to build and actively manage a portfolio of drugs that addresses unmet medical needs and create value in patient therapy.
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, pre-clinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. Our commercial opportunity will be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer side effects or are less expensive than products that we may develop. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies and technology licenses complementary to our programs or advantageous to our business.
We expect that our ability to compete effectively will depend upon our ability to:
|
●
|
successfully and rapidly complete adequate and well-controlled clinical trials that demonstrate statistically significant safety and efficacy and to obtain all requisite regulatory approvals in a cost-effective manner;
|
|
●
|
maintain a proprietary position for our manufacturing processes and other technology;
|
|
●
|
attract and retain key personnel; and
|
|
●
|
build an adequate sales and marketing infrastructure for any approved products.
|
Failure to do one or more of these activities could have an adverse effect on our business, financial condition or results of operations.
Employees
We have four full-time employees and retain the services of approximately 19 persons on an independent contractor/consultant and contract-employment or full-time employee basis. As such, we currently operate in a “virtual” corporate structure in order to minimize fixed personnel costs. Over time, we plan to establish a base of full time employees and corporate infrastructure.
We are not party to any material legal proceedings.
Our corporate headquarters are located at Suite 720-999 West Broadway, Vancouver, British Columbia, Canada. Our clinical operations are managed at 3475 Edison Way, Suite R, Menlo Park, California, 94025. Our current monthly base rent for our corporate headquarters is $2,185 (Cdn $2,325) under a one-year lease expiring in November 2014. In addition, Valent, which is owned by Dr. Dennis Brown, our Chief Scientific Officer, leases facilities in California and we have access to such facilities pursuant to an informal unwritten arrangement with Valent. Our leased premises, academic relationships, and access to the Valent facility are sufficient to meet the immediate needs of our business, research and operations.
The Company’s common stock is quoted on the Over-the-Counter Bulletin Board, or OTCBB, under the symbol “DMPI.”
There was no reported trading in our common stock prior to January 25, 2013. Since January 25, 2013, there has been limited trading in our common stock. The following table sets forth the range of high and low bid prices of our common stock as reported and summarized on the OTCQB for the periods indicated. These prices are based on inter-dealer bid and asked prices, without markup, markdown, commissions, or adjustments and may not represent actual transactions.
|
Calendar Quarter
|
|
High Bid
|
|
|
Low Bid
|
|
|
2013 First Quarter
|
|
$
|
2.50
|
|
|
$
|
1.30
|
|
|
2013 Second Quarter
|
|
$
|
2.48
|
|
|
$
|
1.55
|
|
|
2013 Third Quarter
|
|
$
|
2.04
|
|
|
$
|
0.90
|
|
|
2013 Fourth Quarter
|
|
$
|
1.48
|
|
|
$
|
0.75
|
|
|
2014 First Quarter
|
|
$
|
1.60
|
|
|
$
|
0.79
|
|
Trades in our common stock may be subject to Rule 15g-9 of the Exchange Act, which imposes requirements on broker/dealers who sell securities subject to the rule to persons other than established customers and accredited investors. For transactions covered by the rule, broker/dealers must make a special suitability determination for purchasers of the securities and receive the purchaser’s written agreement to the transaction before the sale.
The SEC also has rules that regulate broker/dealer practices in connection with transactions in “penny stocks.” Penny stocks generally are equity securities with a price of less than $5.00 (other than securities listed on certain national exchanges, provided that the current price and volume information with respect to transactions in that security is provided by the applicable exchange or system). The penny stock rules require a broker/dealer, before effecting a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document prepared by the SEC that provides information about penny stocks and the nature and level of risks in the penny stock market. The broker/dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker/dealer and its salesperson in the transaction, and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid and offer quotations, and the broker/dealer and salesperson compensation information, must be given to the customer orally or in writing before effecting the transaction, and must be given to the customer in writing before or with the customer’s confirmation. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for shares of our common stock. As a result of these rules, investors may find it difficult to sell their shares.
The Company has never declared or paid any cash dividends on its common stock. The Company currently intends to retain future earnings, if any, to finance the expansion of its business. As a result, the Company does not anticipate paying any cash dividends in the foreseeable future.
As of June 6, 2014, we have 28,947,760 shares of common stock, 7,044,583 shares of common stock issuable upon exchange of the Exchangeable Shares, warrants to purchase 18,732,485, including 9,195,478 Investor Warrants, shares of common stock, and options to purchase 3,240,000 shares of common stock, issued and outstanding.
As of June 6, 2014, there were approximately 183 holders of record of the Company’s common stock.
Securities Authorized for Issuance Under Equity Compensation Plans
The following table sets forth the aggregate information of our equity compensation plans in effect as of December 31, 2013:
|
Plan
|
|
Number of
securities to be
issued upon exercise
of outstanding
options
and rights
|
|
|
Weighted-average
exercise price of
outstanding options and rights
|
|
|
Number of securities
remaining available for
future issuance under
equity compensation plans
(excluding securities
reflected
in first column
|
|
|
Equity compensation plans approved by security holders
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Equity compensation plans not approved by security holders – Amended and Restated 2003 Employee Stock Option Plan
|
|
|
3,240,000
|
|
|
|
0.96
|
|
|
|
1,069,862
|
|
|
Totals
|
|
|
3,240,000
|
|
|
|
|
|
|
|
1,069,862
|
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS FOR THE FISCAL YEAR ENDED DECEMBER 31, 2013
This Management Discussion and Analysis (“MD&A”) contains “forward-looking statements”, which represent our projections, estimates, expectations or beliefs concerning among other things, financial items that relate to management’s future plans or objectives or to our future economic and financial performance. In some cases, you can identify these statements by terminology such as “may”, “should”, “plans”, “believe”, “will”, “anticipate”, “estimate”, “expect” “project”, or “intend”, including their opposites or similar phrases or expressions. You should be aware that these statements are projections or estimates as to future events and are subject to a number of factors that may tend to influence the accuracy of the statements. These forward-looking statements should not be regarded as a representation by the Company or any other person that the events or plans of the Company will be achieved. You should not unduly rely on these forward-looking statements, which speak only as of the date of this MD&A. Except as may be required under applicable securities laws, we undertake no obligation to publicly revise any forward-looking statement to reflect circumstances or events after the date of this MD&A or to reflect the occurrence of unanticipated events.
You should review the factors and risks we describe under “Risk Factors” included in the Offer to Amend and Exercise and in our Post Effective Amendment No. 1 to our Registration Statement on Form S-1 filed with the SEC on April 17, 2014. Actual results may differ materially from any forward-looking statement.
DelMar Pharmaceuticals, Inc. (the “Company”) is a Nevada corporation formed on June 24, 2009 under the name Berry Only Inc. Prior to the Reverse Acquisition (discussed below), the Company did not have any significant assets or operations. DelMar Pharmaceuticals, Inc. is the parent company of Del Mar Pharmaceuticals (BC) Ltd. (“DelMar (BC)”), a British Columbia, Canada corporation incorporated on April 6, 2010, which is a development stage company with a focus on the development of drugs for the treatment of cancer. The Company is also the parent company to 0959454 B.C. Ltd., a British Columbia corporation (“Callco”), and 0959456 B.C. Ltd., a British Columbia corporation (“Exchangeco”). Callco and Exchangeco were formed to facilitate the Reverse Acquisition.
Pursuant to the Reverse Acquisition, the Company acquired (either directly or indirectly (through Exchangeco)) all of the issued and outstanding shares of DelMar (BC) on January 25, 2013. As a result of the shareholders of DelMar (BC) having a controlling interest in the Company subsequent to the Reverse Acquisition, for accounting purposes the transaction is a capital transaction with DelMar (BC) being the accounting acquirer even though the legal acquirer is Berry. Therefore, the historic financial statements of DelMar (BC) are presented as the comparative balances for the periods prior to the Reverse Acquisition.
Our drug discovery research and development focuses on identifying well-validated clinical and commercial-stage compounds and establishing a scientific rationale for development in modern orphan cancer indications. We conduct further research on promising candidates through our network of consultants and contract research organizations. This approach allows us to identify and advance potential drug candidates without significant investment in “wet lab” infrastructure. Based on this strategy, we acquired intellectual property and prototype drug product related to our lead drug candidate, VAL-083, from Valent Technologies LLC (“Valent”) in September 2010 and initiated new clinical trials in 2011. In addition, we have identified multiple additional drug candidates that we may have the opportunity to license or acquire in the future.
Our lead product candidate, VAL-083, represents a “first in class” small-molecule chemotherapeutic. The molecular structure of VAL-083 is not an analogue or derivative of other small molecule chemotherapeutics approved for the treatment of cancer. VAL-083, which was originally discovered in the 1960’s, has been assessed in multiple clinical studies sponsored by the National Cancer Institute (“NCI”) in the United States as a treatment for various cancers including lung, brain, cervical, ovarian tumors and leukemia. Published pre-clinical and clinical data suggest that VAL-083 may be active against a range of tumor types. VAL-083 is approved as a cancer chemotherapeutic in China for the treatment of chronic myelogenous leukemia (“CML”) and lung cancer. VAL-083 has not been approved for any indications outside of China.
Upon obtaining regulatory approval, we intend to commercialize VAL-083 and other product candidates for the treatment of orphan cancer indications where patients have failed other therapies or have limited medical options. Orphan diseases are defined in the United States under the Rare Disease Act of 2002 as “any disease or condition that affects less than 200,000 persons in the United States”. The Orphan Drug Act of 1983 is a federal law that provides financial and other incentives including a period of market exclusivity to encourage the development of new treatments for orphan diseases.
We research the mechanism of action of our product candidates to determine the clinical indications best suited for therapy and attempt to rapidly advance our product candidates into human clinical trials and toward commercialization.
Central Nervous System Cancers
In October 2011, we initiated clinical trials with VAL-083 as a potential new treatment for glioblastoma multiforme (“GBM”), the most common and aggressive form of brain cancer.
We have presented interim data from our clinical trial at peer-reviewed scientific meetings including the Society for NeuroOncology annual meeting (“SNO” – November, 2012), the American Association of Cancer Research (“AACR” – April 2013), the American Society for Clinical Oncology (“ASCO” – June 2013), and the World Federation of Neuro‐Oncology (“WFNO” – November, 2013). In summary, our interim clinical data supports that VAL-083, at doses tested to date:
|
·
|
Is well tolerated in GBM and secondary-progressive brain tumor patients with no drug-related serious adverse events at doses studied to date;
|
|
·
|
Demonstrates that in dose escalation cohorts 1-3, 25% (2/8) of GBM patients and 17% (1/6) of secondary-progressive brain cancer patients showed stable disease or tumor regression in response to VAL-083 treatment at the doses tested to date. These patients had failed prior therapy. The doses tested in these cohorts were well below those used in historical clinical studies;
|
|
·
|
Discloses that Cohort 3 was expanded to gather additional data on central nervous system (“CNS”) metastatic patients at the 5mg/m2 dose level;
|
|
·
|
Demonstrates that the maximum tolerated dose (“MTD”) has not been reached after completion of cohort three. Continued dose escalation is planned; and
|
|
·
|
Shows a dose-dependent increase in plasma exposure following doses of VAL-083.
|
These data support the further development of VAL-083.
In July 2013 the Company announced the opening of its third clinical trial site at the Brain Tumor Center at University of California, San Francisco (“UCSF”) and in August 2013 the Company received a notice of allowance from the United States Food and Drug Administration (“FDA”) enabling the Company to implement a more rapid dose-escalation scheme in our GBM study. The revised dosing regimen was allowed by the FDA following an extensive safety review of patients treated to date. In comparison to the original dose-escalation scheme, the revised plan will enable the trial to reach higher doses and complete the dose-escalation portion of the clinical trial more quickly by skipping two interim doses.
A summary of our completed and proposed dose escalation scheme, as revised, is as follows:
|
Dose Escalation Scheme (mg/m2)
|
|
|
|
|
|
|
Original
|
|
|
Revised
|
|
|
Patients Treated
|
|
Status
|
| |
1.5 |
|
|
|
1.5 |
|
|
|
3 |
|
Completed – No DLT
|
| |
3.0 |
|
|
|
3.0 |
|
|
|
4 |
* |
Completed – No DLT
|
| |
5.0 |
|
|
|
5.0 |
|
|
|
10 |
* |
Completed – No DLT
|
| |
10.0 |
|
|
|
10.0 |
|
|
|
3 |
|
Completed – No DLT
|
| |
15.0 |
|
|
|
|
|
|
|
|
|
|
| |
20.0 |
|
|
|
20.0 |
|
|
|
3 |
|
Completed – No DLT
|
| |
25.0 |
|
|
|
|
|
|
|
|
|
|
| |
30.0 |
|
|
|
30.0 |
|
|
|
3 |
|
Initiated Feb. 2014
|
|
n.a
|
|
|
|
40.0 |
|
|
3
(planned)
|
|
To be initiated subject to no DLT in 30mg/m2 dose cohort
|
|
*Cohorts 2 and 3 were expanded to allow for patient demand and to gather additional data on CNS metastases patients.
|
During 2014 we plan to continue our clinical trials with VAL-083 as a potential treatment for GBM patients who have failed other therapies. Currently, there is no approved therapy for these patients. The goal of the current trial is to establish a modernized dosing regimen for advancement into registration directed trials in the United States as a potential new therapy for the treatment of refractory GBM.
The activity of VAL-083 against solid tumors, including lung cancer, has been established in both pre-clinical and human clinical trials conducted by the NCI. Lung cancer is characterized as small cell and non-small cell lung cancer (“NSCLC”). NSCLC is the most common type of lung cancer. VAL-083 has demonstrated activity against NSCLC in laboratory studies. VAL-083 was also investigated in a number of clinical trials in the United States and Europe during the 1970s both as a stand-alone therapy and in combination with other chemotherapeutic regimens. VAL-083 has been approved by the Chinese Food and Drug Administration (“CFDA”) (formerly the State Food and Drug Administration) for the treatment of lung cancer in China. However, we believe that the use of the drug in the modern era has been limited by a preference for targeted therapies.
In November 2013, we presented non-clinical data which supports the potential utility of VAL-083 in the context of modern lung cancer therapy at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics Annual Meeting.
We plan to establish a strong scientific and clinical rationale to support out-licensing activities to unlock the potential value of the drug in partnership with larger pharmaceutical companies with the resources and commercial infrastructure to effectively develop and launch a lung cancer product.
Additional Orphan Drug Indications
We have established a high-level scientific rationale for the development of VAL-083 in additional high-value orphan cancer indications. Hematologic cancers such as chronic myelogenous leukemia (“CML”), acute myeloid leukemia (“AML”) are of particular interest based on published human clinical data and lack of effective therapeutic options. We have initiated preliminary discussions with leading cancer researchers regarding the development of a clinical strategy for the development of VAL-083 in hematologic cancers.
In addition to our clinical development activities in the United States, we have obtained exclusive commercial rights to VAL-083 in China. In October 2012, we announced that we had entered into a collaboration agreement with the only manufacturer licensed by the Chinese State Food and Drug Administration to produce the product for the China market. This agreement provides us with certain exclusive commercial rights related to drug supply, which positions us with the potential to generate near-term revenue through product sales or royalties for its approved indications in China while we seek global approval in new indications. Our strategy in China is to develop new clinical and non-clinical data in collaboration with leading cancer researchers to demonstrate the utility of VAL-083 in the treatment of CML and lung cancer, particularly for patients who do not respond to, or cannot access, modern treatments such as tyrosine kinase inhibitors. Management believes the data, if favorable, will allow the repositioning of VAL-083 in the China market, and eventually global markets, for the treatment of hematologic cancers and solid tumors. We anticipate seeking a marketing partner for VAL-083 in China in order to obtain royalty revenue from that market.
We have filed a broad portfolio of new patent applications to protect our intellectual property. Our patent applications claim compositions and methods related to the use of VAL-083 and related compounds as well as methods of synthesis and quality controls for the manufacturing process of VAL-083. In July 2013, our first patent in the United States claiming methods of synthesis for VAL-083 was issued by the United States Patent Office. We continue to prosecute patent cases in the United States and international jurisdictions.
In addition to new patent filings, we intend to seek orphan drug protection and other statutory protection for our intellectual property. In February, 2012, we announced that VAL-083 has been granted Orphan Drug protection for the treatment of glioma, including GBM by the FDA in the United States. In January 2013, the European Medicines Association (“EMA”) granted Orphan Drug protection to VAL-083. The orphan drug designation means that we may sell VAL-083 as a treatment for GBM without competition for seven years in the United States and for ten years in the European Union following market approval, in respect of a medicinal product containing a similar active substance for the same indication.
Drugs granted orphan drug protection generally follow the same regulatory development path as any other pharmaceutical product. However, incentives such as scientific advice and reduction or waiver of registration fees and access to specialized grant funding may be available to support and accelerate development of orphan drug candidates.
Developing Partnerships with Pharmaceutical Companies
Guangxi Wuzhou Pharmaceutical Company
We have a strategic collaboration with Guangxi Wuzhou Pharmaceutical Company (“Guangxi Wuzhou Pharmaceuticals”), a subsidiary of publicly traded Guangxi Wuzhou Zhongheng Group Co., Ltd for the development of VAL-083 (marketed as “DAG” in China). Guangxi Wuzhou Pharmaceuticals has received regulatory approval by the CFDA to manufacture and sell VAL-083 in China as a cancer chemotherapy for the treatment of CML and lung cancer.
We are party to a memorandum of understanding and collaboration agreement, dated October 25, 2012 (the “Guangxi Agreement”), with Guangxi Wuzhou Pharmaceuticals. Pursuant to the Guangxi Agreement, we granted to Guangxi Wuzhou Pharmaceuticals a royalty-free license to certain of our intellectual property, as it relates to quality control and drug production methods for VAL-083, and we agreed that Guangxi Wuzhou Pharmaceuticals will be our exclusive supplier of VAL-083 for clinical trials and sales for the China, United States, Canadian and European markets, subject to Guangxi Wuzhou Pharmaceuticals obtaining and maintaining cGMP certification by the FDA, EMEA or other applicable regulatory agencies, and Guangxi Wuzhou Pharmaceuticals being able to meet volumes ordered by us. Guangxi Wuzhou Pharmaceuticals agreed that it may not sell VAL-083 for markets outside of China to any other purchaser other than us. In addition, Guangxi Wuzhou Pharmaceuticals granted us a pre-emptive right (subject to our acceptance of proposed sales volume and prices) to purchase VAL-083 produced by Guangxi Wuzhou Pharmaceuticals. The collaboration under the Guangxi Agreement establishes an exclusive supply relationship between us and Guangxi Wuzhou Pharmaceuticals to include the Chinese market and all markets outside China. DelMar and Guangxi Wuzhou Pharmaceuticals will work together to ensure the product specifications meet global standards in order to accelerate international development and regulatory approval. Subject to meeting and maintaining cGMP certification, Guangxi Wuzhou Pharmaceuticals will be our exclusive supplier of DAG for injection for clinical development and commercial sales.
The Company and Guangxi Wuzhou Pharmaceuticals plan to develop new clinical data to expand the market in China and to seek regulatory approval for the drug in multiple indications on a global basis. The companies have formed a clinical advisory board to oversee clinical studies. Guangxi Wuzhou Pharmaceuticals will provide funding support for clinical trials conducted in China and we will be responsible for development and commercialization. DelMar is currently seeking to establish a separate collaboration for the distribution, sales and marketing of VAL-083 in China.
The term of the Guangxi Agreement (except as it relates to the exclusive rights in the China market) is indefinite, subject to termination upon written agreement of all parties, or if either party breaches any material term and fails to remedy such breach within 30 days of receipt of notice of the breach, or if any action to be taken thereunder is not agreed to by both parties, provided that such matter is referred to the chief executive officer of both parties, and they are unable to resolve such matter within 90 days. No payments have been made to date under the Guangxi Agreement.
The protection of intellectual property rights in China (where VAL-083 is manufactured pursuant to the Guangxi Agreement with the only manufacturer presently licensed by the SDFA to produce the product for the China market, and where VAL-03 is approved for the treatment of CML and lung cancer) is relatively weak compared to the United States, which may negatively affect our ability to generate revenue from VAL-083.
On January 25, 2013 (the “Closing Date”), the Company entered into and closed an exchange agreement (the “Exchange Agreement”), with DelMar (BC), Callco, Exchangeco, and the securityholders of DelMar (BC). Pursuant to the Exchange Agreement, (i) the Company issued 4,340,417 shares of common stock (the “Parent Shares”) to the shareholders of DelMar (BC) who are United States residents (the “U.S. Holders”) in exchange for the transfer to Exchangeco of all 4,340,417 outstanding common shares of DelMar (BC) held by the U.S. Holders, (ii) the shareholders of DelMar (BC) who are Canadian residents (the “Canadian Holders”) received, in exchange for the transfer to Exchangeco of all 8,729,583 outstanding common shares of DelMar (BC) held by the Canadian Holders, 8,729,583 exchangeable shares (the “Exchangeable Shares”) of Exchangeco, and (iii) outstanding warrants to purchase 3,360,000 common shares of DelMar (BC) and outstanding options to purchase 1,020,000 common shares of DelMar (BC) were deemed to be amended such that, rather than entitling the holder to acquire common shares of DelMar (BC), such options and warrants will entitle the holders to acquire shares of common stock of the Company. The Canadian Holders will be entitled to require Exchangeco to redeem (or, at the option of the Company or Callco, to have the Company or Callco purchase) the Exchangeable Shares, and upon such redemption or purchase to receive an equal number of shares of common stock of the Company. The aggregate of 13,070,000 shares of common stock of the Company issued to the former shareholders of DelMar (BC) (on an as-exchanged basis with respect to the Exchangeable Shares) represents 80.1% of the outstanding shares of common stock of the Company following the closing of the Exchange Agreement (the “Reverse Acquisition”).
Upon completion of the Reverse Acquisition DelMar (BC) became a wholly-owned subsidiary of the Company. As a result of the shareholders of DelMar (BC) having a controlling interest in the Company subsequent to the Reverse Acquisition, for accounting purposes the transaction is a capital transaction with DelMar (BC) being the accounting acquirer even though the legal acquirer is Berry. No goodwill is recorded with respect to the transaction as it does not constitute a business combination. For accounting purposes, the transaction is reflected as a recapitalization of DelMar (BC) and consideration for the Reverse Acquisition was deemed to be the fair value of the shares that were issued by DelMar (BC) to acquire the net liabilities of Berry on January 25, 2013. The net identifiable liabilities of Berry on the Closing Date of the Reverse Acquisition were as follows:
| |
|
$
|
|
| |
|
|
|
|
net liabilities (derivative liability)
|
|
|
2,041,680
|
|
The Company determined the fair value of the shares issued on the Reverse Acquisition to be $1,690,004. As a result of the Reverse Acquisition being treated as a recapitalization of DelMar (BC) the Company recognized the loss of $3,731,684 incurred upon the closing of the Reverse Acquisition as an adjustment to opening deficit in the consolidated statement of stockholder’s deficiency at December 31, 2013.
In connection with the Reverse Acquisition, on January 25, 2013, January 31, 2013, February 8, 2013, February 21, 2013, February 28, 2013, March 1, 2013, and March 6, 2013, the Company entered into and closed a series of subscription agreements with accredited investors (the “Investors”), pursuant to which the Company issued an aggregate of 13,125,002 Units at a purchase price of $0.80 per Unit, for aggregate gross proceeds of $10,500,000 (the “Private Offering”). Each Unit consists of one share of common stock and one five-year warrant (the “Investor Warrants”) to purchase one share of common stock at an exercise price of $0.80. The exercise price of the Investor Warrants is subject to adjustment in the event that the Company sells common stock at a price lower than the exercise price, subject to certain exceptions. The Investor Warrants are redeemable by the Company at a price of $0.001 per Investor Warrant at any time subject to the conditions that (i) the Company’s common stock has traded for twenty (20) consecutive trading days with a closing price of at least $1.60 per share with an average trading volume of 50,000 shares per day and (ii) the underlying shares of common stock are registered.
The Company retained Charles Vista, LLC (the “Placement Agent”) as the Placement Agent for the Private Offering. The Company paid the Placement Agent a cash fee of $1,050,000 (equal to 10% of the gross proceeds), a non-accountable expense allowance of $315,000 (equal to 3% of the gross proceeds), and a one-year consulting fee of $60,000. In addition, the Company incurred other closing costs of approximately $500,000 resulting in net proceeds to the Company of $8,575,000. Certain of the additional closing costs were not eligible to be treated as share issue costs and as a result they have been expensed. Net unit proceeds per the consolidated statements of cash flows include gross unit proceeds less cash issue costs attributable to the common stock only. The portion of the unit issue costs attributable to the derivative liability has been expensed.
In addition, the Company issued to the Placement Agent five-year warrants (the “Placement Agent Warrants”) to purchase 5,250,000 shares of common stock (equal to 20% of the shares of common stock (i) included as part of the Units sold in the Private Offering and (ii) issuable upon exercise of the Investor Warrants) at an exercise price of $0.80, exercisable on a cash or cashless basis.
The Company will pay a warrant commission of 5% of the amount of funds raised by an agent upon the exercise of the Investor Warrants following such redemption.
In connection with the Private Offering, the Company entered into a registration rights agreement with the Investors, pursuant to which the Company agreed to file a registration statement (the “Registration Statement”) registering for resale all shares of common stock (a) included in the Units; and (b) issuable upon exercise of the Investor Warrants, no later than 90 days after the completion of the Private Offering (the “Filing Deadline”) and to use commercially reasonable efforts to cause the Registration Statement to become effective within 180 days of the Filing Deadline. The Company agreed to use commercially reasonably efforts to keep the Registration Statement effective while the Investor Warrants are outstanding.
Certain of the Private Offering costs were incurred by the Company prior to December 31, 2012. These costs of $90,771 were treated as issue costs during the year ended December 31, 2013.
The Company acquired its VAL-083 prototype drug, patents and technology rights from Valent. In addition, Valent incurred a significant portion of the Company’s clinical expenses during the periods ended December 31, 2011 and 2012 and has in turn invoiced the Company for those expenses. One of the Company’s officers and directors is also a Principal of Valent and as result Valent is a related party to the Company.
The following related party transactions and balances have been recorded by the Company.
During the year ended December 31, 2013
The Company paid total cash compensation to its officers of $454,549 for the twelve months ended December 31, 2013.
Included in accounts payable at December 31, 2013 is an aggregate amount owing of $74,754 to the Company’s officers and directors for fees and expenses. The Company pays related party payables incurred for fees and expenses under normal commercial terms.
Included in related party payables at December 31, 2013 is an amount of $44,007 relating to clinical development costs incurred by Valent on behalf of the Company. Additionally, the Company also has a loan payable of $272,372, including accrued interest of $22,372, due to Valent (note 4). One of the directors and officers of the Company is also a Principal of Valent. As a result of the Company not expecting to repay Valent within the next twelve months, the balance of the loan and accrued interest has been disclosed as a long-term liability.
On January 25, 2013, in connection with the Reverse Acquisition (note 3), Valent was issued 1,150,000 shares of common stock of the Company in exchange for Valent reducing certain future royalties under the Assignment Agreement (note 8(g)). As a result of the share issuance the Company has recognized an expense of $598,000 for the year ended September 30, 2013.
The Company granted an aggregate 1,410,000 stock options at an exercise price of $1.05 to its officers and directors.
The Company recognized $44,333 in directors’ fees.
During the year ended December 31, 2012
Pursuant to consulting agreements with the Company’s officers and directors the Company paid a total of $27,022 (CDN $27,000) per month to its officers and directors during the year. Under two of these agreements the directors have elected to receive a portion of their aggregate compensation in the form of units. The Company issued 360,000 units for a total amount of $180,144. The units issued relate to an amount of $15,012 (CDN $15,000) per month from January to December 2012 inclusive. All of the units were issued in February 2012. The Company has recognized $180,144 in services for the year ended December 31, 2012. Of the $180,144, $60,389 has been recognized as general and administrative and $119,755 has been recognized as research and development.
Additionally, under the consulting agreements the Company has paid its officers and directors cash compensation totaling an aggregate $12,006 (CDN $12,000) per month. An amount of $144,072 (CDN $144,000) has been paid in cash to the two individuals for the year ended December 31, 2012.
Included in related party payables at December 31, 2012 is an aggregate amount owing of $133,658 to the Company’s directors in relation to their respective consulting agreements and for reimbursable expenses.
Also included in related party payables December 31, 2012 is an amount of $314,119 relating to clinical development costs incurred by Valent on behalf of the Company. On April 30, 2012, Valent was issued 500,000 common shares for partial settlement of the Company’s accounts payable balance with Valent. The total settlement amount was $253,050. Additionally, the Company has a loan payable, including accrued interest, of $264,352 due to Valent (note 4). One of the directors and officers of the Company is also a Principal of Valent.
Through a Company owned by one of the Company’s directors, a $25,000 retainer was paid pursuant to the unit financing completed by the Company (note 8). The $25,000 is included in accounts payable at December 31, 2012.
The Company granted an aggregate 450,000 stock options at an exercise price of $0.47 (CDN $0.50) to its directors.
The Company transferred a total of 1,390,625 shares from the DelMar Employee Share Purchase Trust to the Company’s directors.
During the year ended December 31, 2011
Pursuant to consulting agreements dated August 1, 2011 with the Company’s officers and directors the Company agreed to compensate its officers and directors for services rendered to the Company. An aggregate $26,550 (CDN $27,000) per month commencing August 1, 2011 and ending December 31, 2012 will be payable pursuant the consulting agreements. Under the consulting agreements the Company and the respective officer or director have mutually agreed that a portion of the compensation payable under the respective agreement shall be deemed to have been invested in the unit offering of the Company as of October 3, 2011. The units issued under these agreements shall have the same terms as the CDN $0.50 units issued by the Company to subscribers of the offering.
For the period from August 1 to December 31, 2011 $19,028 (CDN $20,000) per month was settled by the Company with units resulting in 150,000 units being issued. Total research and development expenses of $71,355 (CDN $75,000) and general and administrative expenses of $23,785 (CDN $25,000) have been recorded for this issuance of units.
The Company also issued 50,000 units to one of its officers for the settlement of accounts payable in the amount of $23,785 (CDN $25,000). The units were measured at fair value using the valuation estimate consistent with the most recent financing.
Included in related party payables at December 31, 2011 is an aggregate amount owing of $21,028 to two of the Company’s directors.
Also included in related party payable at December 31, 2011 is an amount of $496,932 relating to clinical development costs incurred by Valent on behalf of the Company. The Company also has a loan payable, including accrued interest, of $256,831 due to Valent at December 31, 2011.
The Company has issued stock purchase warrants. Based on the terms of certain of these warrants the Company determined that the warrants are a derivative liability which is recognized at fair value at the date of the transaction and re-measured at fair value every reporting period with gains or losses on the changes in fair value recorded in the consolidated statement of loss and comprehensive loss.
The Company issued 4,150,000 units on January 23, 2012, 560,000 on February 27, 2012 and 50,000 on May 10, 2012. In addition, during the year ended December 31, 2011 the Company issued 500,000 units on October 3, 2011, 100,000 on October 7, 2011, and 50,000 on November 11, 2011. In total, the Company issued 5,410,000 units for services in settlement of accounts payable and cash proceeds for an aggregate of $2,671,923 (CDN $2,705,000).
The proceeds from the issuance of 3,000,000 of these units were held in escrow pursuant to an exclusive option investment agreement with a strategic investor. Subsequently, the Company elected to let the option expire and the related units were cancelled and the funds returned from escrow to the subscriber in order for the Company to retain control over certain intellectual property and commercial rights.
During the year ended December 31, 2013, 221,000 warrants were exercised for no additional consideration for 221,000 shares of common stock. As a result, $241,715 of the derivative liability has been reclassified to equity. The warrants that have been exercised were revalued at their exercise date and then the reclassification to equity was recorded.
In connection with the Reverse Acquisition, on January 25, 2013, January 31, 2013, February 8, 2013, February 21, 2013, February 28, 2013, March 1, 2013, and March 6, 2013, the Company entered into and closed a series of subscription agreements with accredited investors (the “Investors”), pursuant to which the Company issued an aggregate of 13,125,002 Units at a purchase price of $0.80 per Unit, for aggregate gross proceeds of $10,500,000 (the “Private Offering”). Each Unit consists of one share of common stock and one five-year warrant (the “Investor Warrants”) to purchase one share of common stock at an exercise price of $0.80. The exercise price of the Investor Warrants is subject to adjustment in the event that the Company sells common stock at a price lower than the exercise price, subject to certain exceptions. The Investor Warrants are redeemable by the Company at a price of $0.001 per Investor Warrant at any time subject to the conditions that (i) the Company’s common stock has traded for twenty (20) consecutive trading days with a closing price of at least $1.60 per share with an average trading volume of 50,000 shares per day and (ii) the underlying shares of common stock are registered.
As a result of the Reverse Acquisition, certain warrants that Berry issued pursuant to a warrant dividend became warrants of the Company (the “Dividend Warrants”). The Dividend Warrants are exercisable at $1.25 per share until January 24, 2018. The Dividend Warrants will only be exercisable at such times as the underlying shares of common stock are registered. The Dividend Warrants will be redeemable by the Company at a price of $0.001 per Dividend Warrant at any time commencing 18 months following the date of issuance subject to the conditions that (i) the Company’s common stock has traded for twenty (20) consecutive trading days with a closing price of at least $2.50 per share and (ii) the underlying shares of common stock are registered. Subject to the conditions set forth therein, the Dividend Warrants may be redeemed by the Company upon not less than sixty (60) days nor more than ninety (90) days prior written notice.
Warrants issued for services
During the year ended December 31, 2013 the Company issued 300,000 warrants for services. The warrants were issued on September 12, 2013 and are exercisable on a cashless basis at an exercise price of $1.76 for five years. As of December 31, 2013 all of the warrants have vested. As a result, at December 31, 2013 the Company has recognized $124,020 in the consolidated statement of operations.
The Company’s derivative liability is summarized as follows:
| |
|
December 31,
2013
$
|
|
|
December 31,
2012
$
|
|
| |
|
|
|
|
|
|
|
Opening balance
|
|
|
121,000
|
|
|
|
106,146
|
|
| |
|
|
|
|
|
|
|
|
|
Issuance of units
|
|
|
3,681,372
|
|
|
|
333,356
|
|
|
Dividend Warrant liability acquired on reverse acquisition
|
|
|
2,041,680
|
|
|
|
-
|
|
|
Warrants issued for services
|
|
|
124,020
|
|
|
|
-
|
|
|
Change in fair value of unexercised warrants
|
|
|
(1,324,051
|
)
|
|
|
(318,502
|
)
|
|
Reclassification to equity upon exercise of warrants
|
|
|
(241,715
|
)
|
|
|
-
|
|
| |
|
|
|
|
|
|
|
|
|
Closing balance
|
|
|
4,402,306
|
|
|
|
121,000
|
|
Selected Annual Information
The financial information reported here in has been prepared in accordance with US GAAP. The Company’s functional currency at December 31, 2013 is the USD. The following table represents selected financial information for the Company as of December 31, 2013 and December 31, 2012.
Selected Balance Sheet Data
| |
|
December 31,
2013
$
|
|
|
December 31,
2012
$
|
|
| |
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
4,136,803
|
|
|
|
17,782
|
|
|
Working capital (deficiency)
|
|
|
4,069,261
|
|
|
|
(942,562
|
)
|
|
Total Assets
|
|
|
4,318,748
|
|
|
|
182,830
|
|
|
Derivative liability
|
|
|
4,402,306
|
|
|
|
121,000
|
|
|
Total shareholders’ deficiency
|
|
|
(817,978
|
)
|
|
|
(1,327,914
|
)
|
Selected Statement of Operations Data
| |
|
December 31,
2013
$
|
|
|
December 31,
2012
$
|
|
|
December 31,
2011
$
|
|
|
Period from April 6, 2010 (inception) to December 31, 2013
$
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
2,342,654
|
|
|
|
1,550,490
|
|
|
|
1,051,139
|
|
|
|
4,985,940
|
|
|
General and administrative
|
|
|
3,952,307
|
|
|
|
1,154,604
|
|
|
|
241,802
|
|
|
|
5,416,312
|
|
|
Change in fair value of derivative
|
|
|
(1,324,051
|
)
|
|
|
(318,502
|
)
|
|
|
-
|
|
|
|
(1,642,553
|
)
|
|
Derivative issuance costs
|
|
|
2,713,220
|
|
|
|
24,742
|
|
|
|
-
|
|
|
|
2,737,962
|
|
|
Foreign exchange (gain) loss
|
|
|
3,030
|
|
|
|
(18,492
|
)
|
|
|
18,137
|
|
|
|
2,178
|
|
|
Shares issued for Valent royalty reduction
|
|
|
598,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
598,000
|
|
|
Interest expense
|
|
|
8,020
|
|
|
|
7,521
|
|
|
|
21,933
|
|
|
|
37,474
|
|
|
Interest income
|
|
|
(2,491
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
(2,491
|
)
|
|
Loss from operations
|
|
|
8,290,689
|
|
|
|
2,400,363
|
|
|
|
1,333,011
|
|
|
|
12,132,822
|
|
|
Weighted average number of shares outstanding
|
|
|
29,667,324
|
|
|
|
13,232,349
|
|
|
|
8,527,466
|
|
|
|
-
|
|
|
Loss per share
|
|
|
(0.28
|
)
|
|
|
(0.18
|
)
|
|
|
(0.16
|
)
|
|
|
-
|
|
Expenses net of share-based payments
| |
|
|
|
|
|
|
The following table discloses research and development, and general and administrative expenses net of share-based payment expenses.
|
| |
|
December 31,
2013
$
|
|
|
December 31,
2012
$
|
|
|
December 31,
2011
$
|
|
| |
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
2,342,654
|
|
|
|
1,550,490
|
|
|
|
1,051,139
|
|
|
Share-based payments included in research and development
|
|
|
(568,725
|
)
|
|
|
(866,111
|
)
|
|
|
(232,142
|
)
|
|
Research and development net of share-based compensation
|
|
|
1,773,929
|
|
|
|
684,379
|
|
|
|
818,997
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
General and administrative
|
|
|
3,952,307
|
|
|
|
1,154,604
|
|
|
|
241,802
|
|
|
Share-based payments included in general and administrative
|
|
|
(1,702,061
|
)
|
|
|
(493,652
|
)
|
|
|
(47,570
|
)
|
|
General and administrative net of share-based compensation
|
|
|
2,250,246
|
|
|
|
660,952
|
|
|
|
194,232
|
|
Comparison of the year ended December 31, 2013 and 2012
| |
|
Year Ended
|
|
|
|
|
| |
|
December 31,
2013
$
|
|
|
December 31
2012
$
|
|
|
Change
$
|
|
Change
%
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
2,342,654
|
|
|
|
1,550,490
|
|
|
|
792,164
|
|
|
|
51
|
|
|
General and administrative
|
|
|
3,952,307
|
|
|
|
1,154,604
|
|
|
|
2,797,703
|
|
|
|
242
|
|
|
Change in fair value of derivative liability
|
|
|
(1,324,051
|
)
|
|
|
(318,502
|
)
|
|
|
(1,005,549
|
)
|
|
|
316
|
|
|
Shares issued to Valent for future royalty reduction
|
|
|
598,000
|
|
|
|
-
|
|
|
|
598,000
|
|
|
|
100
|
|
|
Derivative issue costs
|
|
|
2,713,220
|
|
|
|
24,742
|
|
|
|
2,688,478
|
|
|
|
10,866
|
|
|
Foreign exchange (gain) loss
|
|
|
3,030
|
|
|
|
(18,492
|
)
|
|
|
21,522
|
|
|
|
(116)
|
|
|
Interest expense
|
|
|
8,020
|
|
|
|
7,521
|
|
|
|
499
|
|
|
|
7
|
|
|
Interest income
|
|
|
(2,491
|
)
|
|
|
-
|
|
|
|
(2,491
|
)
|
|
|
100
|
|
|
Net loss
|
|
|
8,290,689
|
|
|
|
2,400,363
|
|
|
|
5,890,326
|
|
|
|
|
|
Research and development expenses increased to $2,342,654 for the year ended December 31, 2013 from $1,550,490 for the year ended December 31, 2012. Share-based payments attributable to research and development were $568,725 in the year ended December 31, 2013 compared to $866,111 for the year ended December 31, 2012. In regards to research and development expenses during the year ended December 31, 2013 the Company incurred share-based payments relating to stock options and the issuance of shares for services. For the year ended December 31, 2012 the Company recognized the fair value of shares issued from the DelMar Employee Share Purchase Trust (the “Trust”) to employees and consultants for services rendered to the Company, stock option expense as the Company’s first grant of stock options occurred in February 2012, and the fair value amount recognized for units issued for services. All of the shares had been issued from the Trust at December 31, 2012 and as a result no additional expense was recognized during the year ended December 31, 2013.
After considering the impact of share-based payments research and development expenses increased in the year ended December 31, 2013 to $1,773,929 from $684,379 for the year ended December 31, 2012. The largest component of research and development for the year ended December 31, 2013 was clinical development costs as the Company continued with its Phase I/II clinical trial with VAL-083. The clinical development costs were higher in the current period compared to the prior period largely due to the timing of patient enrollment. Additionally, personnel, intellectual property, and travel costs were all higher during the year ended December 31, 2013 compared to the year ended December 31, 2012.
Personnel costs have increased due to the officers and directors of the Company being compensated with cash during the year ended December 31, 2013 while during the year ended December 31, 2012 a portion of management compensation was in the form of units. Intellectual property costs have increased in the current year as a result of the Company becoming more active in filing and advancing its patents compared to the prior year. Travel has increased in 2013 compared to 2012 as a result of increased travel to scientific and medical conferences to present data and meet with potential collaborators.
General and Administrative
General and administrative expenses were $3,952,307 for the year ended December 31, 2013 compared to $1,154,604 for the year ended December 31, 2012. The increase was partially attributable to an increase in share-based payments to $1,702,061 for the year ended December 31, 2013 compared to $493,652 for the year ended December 31, 2012. In relation to general and administrative expenses during the year ended December 31, 2013 the Company incurred share-based payments relating to stock options, shares issued for services, and warrants issued for services. For the year ended December 31, 2012 the Company recognized the fair value of shares issued from the Trust to employees and consultants for services rendered to the Company, stock option expense as the Company’s first grant of stock options occurred in February 2012, and the fair value amount recognized for warrants and units issued for services. All of the shares had been issued from the Trust at December 31, 2012 and as a result no additional expense was recognized during the year ended December 31, 2013.
After considering the impact of share-based payments, general and administrative expenses increased in the year ended December 31, 2013 to $2,250,246 from $660,952 for the year ended December 31, 2012.
The principal reason for the increase was due to professional fees related to the Company’s Reverse Acquisition and the preparation and filing of the Company’s Registration Statement on Form S-1. A significant portion of the accounting and legal fees related to the Reverse Acquisition were expensed as they did not quality to be recognized as direct share issue costs. The fees and expenses for professional fees for the Reverse Acquisition and the S-1 were one-time fess that will not be incurred in subsequent periods. Additionally, as a result of the Company becoming public due to its Reverse Acquisition, the Company has incurred investor relations fees which it did not incur during the year ended December 31, 2012. The Company becoming a public reporting entity has also led to higher travel costs due to the need to attend more investor and business development conferences.
Personnel, and office and sundry increased in the current year compared to the prior year. Personnel costs have increased due to the officers and directors being compensated with cash in the year ended December 31, 2013 while in the year ended December 31, 2012 a portion of management compensation was in the form of units. In addition, as a result of the Company becoming a public entity, additional officers joined the Company during 2013. Office and sundry increased for the year ended December 31, 2013 compared to the year ended December 31, 2012 largely due an increase in filing and related fees. As a result of the Reverse Acquisition the Company become a public company and began filing obligations with various regulatory authorities.
Change in fair value of derivative liability
Based on the terms of certain warrants issued by the Company, the Company determined that the warrants were a derivative liability which is recognized at fair value at the date of the transaction and re-measured at fair value every reporting period with gains or losses on the changes in fair value recorded in the consolidated statement of loss and comprehensive loss. The balance recognized during the year ended December 31, 2013 was due to a reduction in the Company’s share price between the date the warrants were issued and December 31, 2013 which was the revaluation date.
The Company recognized a gain of $1,324,051 from the change in fair value of the derivative liability for the year ended December 31, 2013 compared to a gain of $318,502 for the year ended December 31, 2012. Changes in the Company’s common stock price can result in significant volatility in the Company’s reported net loss due to its impact on the fair value of the derivative liability. As a result of revaluation gains and losses, it is expected that the Company’s reported net income or loss will continue to experience large fluctuations.
Issuance of Shares to Valent for future royalty reduction
On January 25, 2013, in connection with the Reverse Acquisition, the Company issued to Valent 1,150,000 shares of common stock in exchange for Valent reducing certain future royalties under the Assignment Agreement. As a result of the share issuance the Company has recognized an expense of $598,000 for the year ended December 31, 2013.
The proceeds from the $0.80 unit offering have been allocated between common stock and derivative liability based on the respective fair values of the shares of common stock and the warrants on the issuance date. Additionally, the unit issue costs have also been allocated between common stock and derivative liability on the same pro rata basis as the proceeds. The portion of the issue costs allocated to the derivative liability has been expensed in the consolidated statement of loss and comprehensive loss. The Company recognized $2,713,220 in derivative issue costs for the year ended December 31, 2013. Derivative issue costs of $24,742 related to the issuance of the CDN $0.50 units were recognized for the year ended December 31, 2012.
The Company’s functional currency at December 31, 2013 is the USD but the Company incurs a portion of its expenses in CDN. The foreign exchange gains and losses are reported in other (income) loss in the consolidated statement of loss and comprehensive loss.
The Company recognized a foreign exchange loss of $3,030 for the year ended December 31, 2013 compared to a gain of $18,492 for the year ended December 31, 2012. The change was due to changes in the exchange rate between the CDN and the USD and to varying levels of CDN cash and accounts payable.
Pursuant to a loan agreement dated February 3, 2011, the Company has received a loan from Valent in the amount of $250,000 for the purchase of the prototype drug product. The loan is payable on demand, unsecured and bears interest at 3.00% per year. As a result of the loan payable the Company recognized $8,020 and $7,521 respectively in accrued interest for the years ended December 31, 2013 and 2012.
Year Ended December 31, 2012 compared to the year ended December 31, 2011
| |
|
December 31,
2012
$
|
|
|
December 31,
2011
$
|
|
|
Change
$
|
|
|
Change
%
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
1,550,490
|
|
|
|
1,051,139
|
|
|
|
499,351
|
|
|
|
48
|
|
|
General and administrative
|
|
|
1,154,604
|
|
|
|
241,802
|
|
|
|
912,802
|
|
|
|
377
|
|
|
Change in fair value of derivative
|
|
|
(318,502
|
)
|
|
|
-
|
|
|
|
(318,502
|
)
|
|
|
(100
|
)
|
|
Derivative issuance costs
|
|
|
24,742
|
|
|
|
-
|
|
|
|
24,742
|
|
|
|
100
|
|
|
Foreign exchange (gain) loss
|
|
|
(18,492
|
)
|
|
|
18,137
|
|
|
|
(36,629
|
)
|
|
|
(202
|
)
|
|
Interest expense
|
|
|
7,521
|
|
|
|
21,933
|
|
|
|
(14,412
|
)
|
|
|
(66
|
)
|
|
Net loss
|
|
|
2,400,363
|
|
|
|
1,333,011
|
|
|
|
1,067,352
|
|
|
|
80
|
|
Research and development expenses increased to $1,550,490 for the year ended December 31, 2012 from $1,051,139 for the year ended December 31, 2011. The largest component of research and development for the year ended December 31, 2012 was share-based payments. The large increase in share-based payments for the current year compared to the prior year was due to increases in the recognition of the fair value of shares issued from Trust to employees and consultants for services rendered to the Company, stock option expenses as the Company’s first grant of stock options occurred in February 2012, the recognition of the fair value of shares issued for services, and the increase in the fair value amount recognized for units issued for services. In the prior year shares issued from the Trust did not occur until October 2011 and there were no shares issued for services to December 31, 2011 so as a result there were no expenses related to shares for services recognized during the year ended December 31, 2011. Units were issued for services in both periods but for the year ended December 31, 2012 agreements applicable to units issued for services covered the entire year ended December 31, 2012 while in the year ended December 31, 2011 units for services were applicable for only five months resulting in a lower expense in the prior year. At December 31, 2012 all of the shares have been issued from the Trust and the agreements with management for the issuance of units for services have expired. As a result, it is not expected that additional share-based payment expenses for these two items will be incurred in the future.
Additionally, contracted research, personnel, and travel were higher during the year ended December 31, 2012 compared to the year ended December 31, 2011. Contracted research costs were higher in the current year due to the initiation of nonclinical research studies supporting new indications in the current period. There were no such nonclinical studies on-going in the prior period. Travel has increased in the current period compared to the prior period as a result of increased travel to scientific and medical conferences. Personnel costs have increased due to one director receiving cash payments during 2012 while he received share-based payments in 2011. Partially offsetting the impact of higher contracted research, personnel, travel and share-based payments was a reduction in clinical development expenses related to the clinical trials being undertaken with VAL-083 for the year ended December 31, 2012 compared to the year ended December 31, 2011. The clinical development costs were lower in the current year compared to the prior year largely due to clinical preparation and start-up costs incurred in the year ended December 31, 2011 compared to the year ended December 31, 2012. Intellectual property costs have decreased in the current year as a result of $89,432 being recognized during the year ended December 31, 2011 from the fair value of warrants issued to Valent for the transfer of patents and intellectual property rights to the Company.
General and Administrative
General and administrative expenses were $1,154,604 for the year ended December 31, 2012 compared to $241,802 for the year ended December 31, 2011. The principal reasons for the increase were due to higher professional fees, share-based payments, travel, and personnel costs incurred in the current year compared to the prior year. The increase in professional fees related to costs incurred for the initiation of the Company’s first financial statement audit, legal fees related to the updating of the Company’s corporate records, and for business development fees incurred in relation to the Company’s collaboration in China and for activities relating to preparation for the Company’s financing and reverse acquisition transaction that was completed in January 2013. Share-based payments have increased partially due to stock option expenses as the Company’s first grant of stock options occurred in February 2012. Additionally, units were issued for services in both periods but for the year ended December 31, 2012 agreements applicable to units issued for services covered the entire year while in the year ended December 31, 2011 units for services were applicable for only five months resulting in a lower expense in the prior year. At December 31, 2012 all of the shares have been issued from the DelMar Employee Share Purchase Trust and the agreements with management for the issuance of units for services have expired. As a result, it is not expected that additional share-based payment expenses for these two items will be incurred in the future. Travel costs have increased in the current year largely due to expenses associated with preparations for the Company’s financing which was completed in January, 2013. Personnel costs increased in the year ended December 31, 2012 compared to the year ended December 31, 2011 due to an increase in salaries paid in the current year compared to the prior year.
Change in fair value of derivative liability
Based on the terms of the warrants issued as part of the Company’s CDN $0.50 units it was determined that the warrants were considered a derivative liability which is recognized at fair value at the date of the transaction and re-measured at fair value every reporting period with gains or losses on the changes in fair value recorded in the statement of loss and comprehensive loss. The Company recognized a gain of $318,502 from the change in fair value of the derivative liability at December 31, 2012. There was no change in the fair value of the derivative liability for the year ended December 31, 2011.
The proceeds from the CDN $0.50 unit offering have been allocated between common stock and derivative liability based on the fair values of the common shares and the warrants. The portion of the issue costs allocated to the derivative liability has been expensed the statement of loss and comprehensive loss. The Company recognized $24,742 in derivative issue costs at December 31, 2012. There was no derivative issue costs recognized for the year ended December 31, 2011.
Foreign Exchange (Gain) Loss
The Company’s functional currency at December 31, 2012 was the CDN but the Company reported its results in USD. The translation gains and losses are reported in other comprehensive loss. Foreign exchange gains and losses are the result of the Company incurring expenses in USD and then translating those USD expenses into CDN.
The Company recognized a foreign exchange gain of $18,492 for the year ended December 31, 2012 compared to a loss of $18,137 for the year ended December 31, 2011. The change was due to changes in the exchange rate between the CDN and the USD and to varying levels of USD cash and accounts payable.
Pursuant to a loan agreement dated February 3, 2011, the Company has entered a loan with Valent in the amount of $250,000 for the purchase of the prototype drug product. The loan is unsecured and bears interest at 3.00% per year. As a result of the loan payable the Company recognized $7,521 and $6,831 respectively in accrued interest for the years ended December 31, 2012 and 2011. During the year ended December 31, 2011 the Company was charged $15,102 in interest expense relating to outstanding trade payable balances.
Liquidity and Capital Resources
Year ended December 31, 2013 compared to the year ended December 31, 2012
| |
|
December 31,
2013
$
|
|
|
December 31,
2012
$
|
|
|
Change
$
|
|
|
Change
%
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash used in operating activities
|
|
|
(5,520,499
|
)
|
|
|
(578,035
|
)
|
|
|
(4,942,464
|
)
|
|
|
855
|
|
|
Cash flows from financing activities
|
|
|
9,639,520
|
|
|
|
580,799
|
|
|
|
9,058,721
|
|
|
|
1,560
|
|
Net cash used in operating activities increased to $5,520,499 for the year ended December 31, 2013 from $578,035 for the year ended December 31, 2012. The increase was largely the result of an increase in the net loss to $8,290,689 for the year ended December 31, 2013 compared to $2,400,363 for the year ended December 31, 2012. Partially offsetting the impact on cash of the higher net loss were non-cash items totaling $3,753,763 incurred in the current year consisting of accrued loan interest, change in fair value of the derivative liability, warrants issued for services, shares issued to Valent for a future royalty reduction, non-cash derivative issue costs and share-based payments. The non-cash items for the year ended December 31, 2012 totaled $1,048,782 and consisted of accrued loan interest, change in fair value of the derivative liability, units issued for services, warrants issued for services, and share-based payments. The largest changes in non-cash working capital for the year ended December 31, 2013 were outflows of $537,158 and $338,747 from the payment of accounts payable and accrued liabilities, and related party payables respectively. In the year ended December 31, 2012 there was an inflow of $865,007 from an increase accounts payable and accrued liabilities and an outflow of $70,183 from a reduction in related party payables. Additionally, during the year ended December 31, 2013 and 2012 there were respective outflows of $142,105 and $14,581 from increases in prepaid expenses.
As a result of the Company’s expectations as to the timing of the repayment of the Valent loan, the Company has presented the full loan and accrued interest balance as a long-term liability at December 31, 2013 and December 31, 2012.
The Company received $9,639,520 in net proceeds from the issuance of units during the year ended December 31, 2013 compared to $671,570 in net proceeds from the issuance of units during the year ended December 31, 2012. Also in 2012, the Company incurred $90,771 in deferred financing costs that were treated as share issue costs in 2013. The net proceeds from units issued in 2013 were $8,575,000. However, as a result of a portion of the unit proceeds and issue costs being accounted for as a derivative liability the net proceeds on the consolidated statement of cash flows is $9,639,520. During the year ended December 31, 2013 certain of the additional closing costs were not eligible to be treated as share issue costs and as a result they have been expensed. Net unit proceeds per the consolidated statements of cash flows include gross unit proceeds less cash issue costs attributable to the shares only. The portion of the unit issue costs attributable to the derivative liability has been expensed.
The units issued in the year ended December 31, 2013 were the $0.80 units issued in conjunction with the Reverse Acquisition while in the prior period the units issued were the CDN $0.50 units.
Operating Capital and Capital Expenditure Requirements
Liquidity and capital resources
For the year ended December 31, 2013, the Company reported a net loss of $8,290,689 and an accumulated deficit of $15,864,506 at that date. As at December 31, 2013, the Company has cash and cash equivalents of $4,136,803 and a working capital balance of $4,069,261. The Company does not have the prospect of achieving any significant revenues in the immediate near future and the Company will require additional funding to maintain its research and development projects and for general operations. There is a large degree of uncertainty as to the expenses the Company will incur in developing and pursuing its business plan. In addition, the Company has not begun to generate revenues from any product candidate.
Consequently, management is pursuing various financing alternatives to fund the Company’s operations so it can continue as a going concern in the medium to longer term. Accordingly, the Company is considered to be in the development stage as defined in Accounting Standards Codification (ASC) 915-10. We believe, based on our current estimates and plans we expect to have enough cash to fund our operations for the next 12 to 15 months. Management plans to secure the necessary financing through the issue of new equity and/or the entering into of strategic partnership arrangements. Nevertheless, there is no assurance that these initiatives will be successful.
These financial statements have been prepared on a going concern basis which assumes that the Company will continue its operations for the foreseeable future and contemplates the realization of assets and the settlement of liabilities in the normal course of business.
The conditions and risks noted above cast substantial doubt on the validity of that assumption. These financial statements do not give effect to any adjustments to the amounts and classification of assets and liabilities that may be necessary and could potentially be material, should the Company be unable to continue as a going concern.
There could be material differences in our cost estimates or there can be unforeseen events, problems or delays will occur that would require us to seek additional debt and/or equity funding. The ability of the Company to meet its obligations and continue the research and development of its product candidate is dependent on its ability to continue to raise adequate financing. There can be no assurance that such financing will be available to the Company in the amount required at any time or for any period or, if available, that it can be obtained on terms satisfactory to the Company. The Company may tailor its drug candidate program based on the amount of funding it raises.
Our future funding requirements will depend on many factors, including but not limited to:
|
·
|
the rate of progress and cost of our clinical trials, preclinical studies and other discovery and research and development activities;
|
|
·
|
the costs associated with establishing manufacturing and commercialization capabilities;
|
|
·
|
the costs of acquiring or investing in businesses, product candidates and technologies;
|
|
·
|
the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights;
|
|
·
|
the costs and timing of seeking and obtaining FDA and other regulatory approvals;
|
|
·
|
the effect of competing technological and market developments; and
|
|
·
|
the economic and other terms and timing of any collaboration, licensing or other arrangements into which we may enter.
|
Until we can generate a sufficient amount of product revenue to finance our cash requirements, which we may never do, we expect to finance future cash needs primarily through public or private equity offerings, debt financings or strategic collaborations. Although we are not reliant on institutional credit finance and therefore not subject to debt covenant compliance requirements or potential withdrawal of credit by banks, the current economic climate has also impacted the availability of funds and activity in equity markets. We do not know whether additional funding will be available on acceptable terms, or at all. If we are not able to secure additional funding when needed, we may have to delay, reduce the scope of our clinical trials or research and development programs or make changes to our operating plan. In addition, we may have to partner our product candidate program at an earlier stage of development, which would lower the economic value of the program to us.
Critical Accounting Policies
The preparation of financial statements, in conformity with generally accepted accounting principles in the United States, requires companies to establish accounting policies and to make estimates that affect both the amount and timing of the recording of assets, liabilities, revenues and expenses. Some of these estimates require judgments about matters that are inherently uncertain and therefore actual results may differ from those estimates.
|
A detailed summary of all of the Company’s significant accountings policies and the estimates derived therefrom is included in Note 2 to the Company’s Financial Statements for the year ended December 31 2013. While all of the significant accounting policies are important to the Company’s consolidated financial statements, the following accounting policies and the estimates derived therefrom have been identified as being critical:
|
| |
|
· Shares for services
|
|
· Stock options
|
|
· Derivative liability
|
The Company has issued equity instruments for services provided by employees and nonemployees. The equity instruments are valued at the fair value of the instrument granted (see notes 7 and 8 of the consolidated financial statements for assumptions).
In prior periods the Company transferred shares from the DelMar Employee Share Purchase Trust (the “Trust”) to consultants and management in exchange for services rendered to the Company. The Company recognizes the fair value of the shares transferred as an expense with a corresponding increase in common stock. The shares reserved for issuance to consultants and management that are held by the Trust are included in the financial statements at year end. There are no other assets in the Trust. The number of shares outstanding for issue from the Trust at December 31, 2013 is nil (December 31, 2012 – nil).
The shares transferred from the Trust in prior periods have been valued using the fair value of the shares transferred. The Company has used recent share transactions in order to determine the fair value of the shares transferred from the Trust.
The Company accounts for these awards under ASC 718, “Compensation - Stock Compensation” (“ASC 718”). ASC 718 requires measurement of compensation cost for all stock-based awards at fair value on the date of grant and recognition of compensation over the requisite service period for awards expected to vest. Compensation expense for unvested options to non-employees is revalued at each period end and is being amortized over the vesting period of the options. The determination of grant-date fair value for stock option awards is estimated using the Black-Scholes model, which includes variables such as the expected volatility of the Company’s share price, the anticipated exercise behavior of its grantee, interest rates, and dividend yields. These variables are projected based on the Company’s historical data, experience, and other factors. Changes in any of these variables could result in material adjustments to the expense recognized for share-based payments. Such value is recognized as expense over the requisite service period, net of estimated forfeitures, using the straight-line attribution method. The estimation of stock awards that will ultimately vest requires judgment, and to the extent actual results or updated estimates differ from current estimates, such amounts are recorded as a cumulative adjustment in the period estimates are revised. The Company considers many factors when estimating expected forfeitures, including type of awards granted, employee class, and historical experience. Actual results and future estimates may differ substantially from current estimates.
The Company accounts for certain warrants under the authoritative guidance on accounting for derivative financial instruments indexed to, and potentially settled in, a company’s own stock, on the understanding that in compliance with applicable securities laws, the warrants require the issuance of securities upon exercise and do not sufficiently preclude an implied right to net cash settlement. The Company classifies warrants in its balance sheet as a derivative liability which is fair valued at each reporting period subsequent to the initial issuance. As quoted prices for the derivative liability are not available, the Company uses a simulated probability valuation model to value the warrants. Determining the appropriate fair-value model and calculating the fair value of warrants requires considerable judgment. Any change in the estimates used may cause the value to be higher or lower than that reported. The estimated volatility of the Company’s common stock at the date of issuance, and at each subsequent reporting period, is based on the historical volatility of similar life sciences companies. The risk-free interest rate is based on rates published by the government for bonds with a maturity similar to the expected remaining life of the warrants at the valuation date. The expected life of the warrants is assumed to be equivalent to their remaining contractual term.
Off-Balance Sheet Arrangements
We do not have any off balance sheet arrangements.
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS FOR THE QUARTER ENDED MARCH 31, 2014
This Management Discussion and Analysis (“MD&A”) contains “forward-looking statements”, which represent our projections, estimates, expectations or beliefs concerning among other things, financial items that relate to management’s future plans or objectives or to our future economic and financial performance. In some cases, you can identify these statements by terminology such as “may”, “should”, “plans”, “believe”, “will”, “anticipate”, “estimate”, “expect” “project”, or “intend”, including their opposites or similar phrases or expressions. You should be aware that these statements are projections or estimates as to future events and are subject to a number of factors that may tend to influence the accuracy of the statements. These forward-looking statements should not be regarded as a representation by the Company or any other person that the events or plans of the Company will be achieved. You should not unduly rely on these forward-looking statements, which speak only as of the date of this MD&A. Except as may be required under applicable securities laws, we undertake no obligation to publicly revise any forward-looking statement to reflect circumstances or events after the date of this MD&A or to reflect the occurrence of unanticipated events.
You should review the factors and risks we describe under “Risk Factors” in Post Effective Amendment No. 1 to our Registration Statement on Form S-1 filed with the SEC on April 17, 2014 and in the Offer to Amend and Exercise. Actual results may differ materially from any forward-looking statement.
DelMar Pharmaceuticals, Inc. (the “Company”) is a Nevada corporation formed on June 24, 2009 under the name Berry Only Inc. Prior to the Reverse Acquisition (discussed below), the Company did not have any significant assets or operations. DelMar Pharmaceuticals, Inc. is the parent company of Del Mar Pharmaceuticals (BC) Ltd. (“DelMar (BC)”), a British Columbia, Canada corporation incorporated on April 6, 2010, which is a development stage company with a focus on the development of drugs for the treatment of cancer. The Company is also the parent company to 0959454 B.C. Ltd., a British Columbia corporation (“Callco”), and 0959456 B.C. Ltd., a British Columbia corporation (“Exchangeco”). Callco and Exchangeco were formed to facilitate the Reverse Acquisition.
Pursuant the Reverse Acquisition, the Company acquired (either directly or indirectly (through Exchangeco)) all of the issued and outstanding shares of DelMar (BC) on January 25, 2013. As a result of the shareholders of DelMar (BC) having a controlling interest in the Company subsequent to the Reverse Acquisition, for accounting purposes the transaction is a capital transaction with DelMar (BC) being the accounting acquirer even though the legal acquirer is Berry. Therefore, the historic financial statements of DelMar (BC) are presented as the comparative balances for the periods prior to the Reverse Acquisition.
References to the Company, “we”, “us”, and “our” refer to the Company and its wholly-owned subsidiaries, DelMar (BC), Callco and Exchangeco. References to Berry relate to the Company prior to the Reverse Acquisition.
Our drug discovery research and development focuses on identifying well-validated clinical and commercial-stage compounds and establishing a scientific rationale for development in modern orphan cancer indications. We conduct further research on promising candidates through our network of consultants and contract research organizations. This approach allows us to identify and advance potential drug candidates without significant investment in “wet lab” infrastructure. Based on this strategy, we acquired intellectual property and prototype drug product related to our lead drug candidate, VAL-083, from Valent Technologies LLC (“Valent”) in September 2010 and initiated new clinical trials in 2011.
For the three months ended March 31, 2014, the Company reported a net loss of $3,198,607 and an accumulated deficit of $19,063,113 at that date. As at March 31, 2014, the Company has cash and cash equivalents of $3,474,150 and a working capital balance of $3,313,942. The Company does not have the prospect of achieving any significant revenues in the immediate near future and the Company will require additional funding to maintain its research and development projects and for general operations. There is a large degree of uncertainty as to the expenses the Company will incur in developing and pursuing its business plan. In addition, the Company has not begun to generate revenues from any product candidate.
Consequently, management is pursuing various financing alternatives to fund the Company’s operations so it can continue as a going concern in the medium to longer term. Accordingly, the Company is considered to be in the development stage as defined in Accounting Standards Codification (ASC) 915-10. We believe, based on our current estimates and plans we expect to have enough cash to fund our operations for the next 9 to 12 months. Management plans to secure the necessary financing through the issuance of new equity and/or the entering into of strategic partnership arrangements. Nevertheless, there is no assurance that these initiatives will be successful.
The Company’s financial statements have been prepared on a going concern basis which assumes that the Company will continue its operations for the foreseeable future and contemplates the realization of assets and the settlement of liabilities in the normal course of business.
The conditions and risks noted above cast substantial doubt on the validity of that assumption. These financial statements do not give effect to any adjustments to the amounts and classification of assets and liabilities that may be necessary and could potentially be material, should the Company be unable to continue as a going concern.
There could be material differences in our cost estimates or there can be unforeseen events, problems or delays will occur that would require us to seek additional debt and/or equity funding. The ability of the Company to meet its obligations and continue the research and development of its product candidate is dependent on its ability to continue to raise adequate financing. There can be no assurance that such financing will be available to the Company in the amount required at any time or for any period or, if available, that it can be obtained on terms satisfactory to the Company. The Company may tailor its drug candidate program based on the amount of funding it raises.
Our lead product candidate, VAL-083, represents a “first in class” small-molecule chemotherapeutic. The molecular structure of VAL-083 is not an analogue or derivative of other small molecule chemotherapeutics approved for the treatment of cancer. VAL-083, which was originally discovered in the 1960’s, has been assessed in multiple clinical studies sponsored by the National Cancer Institute (“NCI”) in the United States as a treatment for various cancers including lung, brain, cervical, ovarian tumors and leukemia. Published pre-clinical and clinical data suggest that VAL-083 may be active against a range of tumor types. VAL-083 is approved as a cancer chemotherapeutic in China for the treatment of chronic myelogenous leukemia (“CML”) and lung cancer. VAL-083 has not been approved for any indications outside of China.
Upon obtaining regulatory approval, we intend to commercialize VAL-083 and other product candidates for the treatment of orphan cancer indications where patients have failed other therapies or have limited medical options. Orphan diseases are defined in the United States under the Rare Disease Act of 2002 as “any disease or condition that affects less than 200,000 persons in the United States”. The Orphan Drug Act of 1983 is a federal law that provides financial and other incentives including a period of market exclusivity to encourage the development of new treatments for orphan diseases.
We research the mechanism of action of our product candidates to determine the clinical indications best suited for therapy and attempt to rapidly advance our product candidates into human clinical trials and toward commercialization.
Central Nervous System Cancers
In October 2011, we initiated clinical trials with VAL-083 as a potential new treatment for glioblastoma multiforme (“GBM”), the most common and aggressive form of brain cancer.
We have presented interim data from our clinical trials at peer-reviewed scientific meetings including the Society for NeuroOncology annual meeting (“SNO” – November, 2012), the American Association of Cancer Research (“AACR” – April 2013), the American Society for Clinical Oncology (“ASCO” – June 2013), the World Federation of NeuroOncology (“WFNO” – November, 2013) and AACR in April 2014. In summary, our interim clinical data support that VAL-083 at doses tested to date indicate that:
|
·
|
VAL-083 is safe and well-tolerated by patients in doses tested to date;
|
|
·
|
A maximum tolerated dose has not yet been reached;
|
|
·
|
A portion of GBM patients who have failed other therapies demonstrate stable disease or tumor regression following treatment with VAL-083 and;
|
|
·
|
Pharmacokinetic analysis demonstrates a dose-dependent plasma exposure.
|
These data support the further development of VAL-083.
In July 2013 the Company announced the opening of its third clinical trial site at the Brain Tumor Center at University of California, San Francisco (“UCSF”) and in August 2013 the Company received a notice of allowance from the United States Food and Drug Administration (“FDA”) enabling the Company to implement a more rapid dose-escalation scheme in our GBM study. The revised dosing regimen was allowed by the FDA following an extensive safety review of patients treated to date. In comparison to the original dose-escalation scheme, the revised plan will enable the trial to reach higher doses and complete the dose-escalation portion of the clinical trial more quickly by skipping two interim doses.
A summary of our completed and proposed dose escalation scheme, as revised, is as follows:
|
Dose Escalation Scheme (mg/m2)
|
|
|
|
|
|
|
Original
|
|
|
Revised
|
|
|
Patients Treated
|
|
Status
|
| |
1.5
|
|
|
|
1.5
|
|
|
|
3
|
|
Completed – No dose limiting toxicity, or “DLT”
|
| |
3.0
|
|
|
|
3.0
|
|
|
|
4
|
*
|
Completed – No DLT
|
| |
5.0
|
|
|
|
5.0
|
|
|
|
10
|
*
|
Completed – No DLT
|
| |
10.0
|
|
|
|
10.0
|
|
|
|
3
|
|
Completed – No DLT
|
| |
15.0
|
|
|
|
|
|
|
|
|
|
|
| |
20.0
|
|
|
|
20.0
|
|
|
|
3
|
|
Completed – No DLT
|
| |
25.0
|
|
|
|
|
|
|
|
|
|
|
| |
30.0
|
|
|
|
30.0
|
|
|
|
3
|
|
Completed – No DLT
|
|
n.a
|
|
|
|
40.0
|
|
|
3
(planned)
|
|
Initiated May 2014
|
|
*Cohorts 2 and 3 were expanded to allow for patient demand and to gather additional data on CNS metastases patients.
|
During 2014 we plan to continue our clinical trials with VAL-083 as a potential treatment for GBM patients who have failed other therapies. Currently, there is no approved therapy for these patients. The goal of the current trial is to establish a modernized dosing regimen for advancement into registration directed trials in the United States as a potential new therapy for the treatment of refractory GBM.
Lung Cancer
The activity of VAL-083 against solid tumors, including lung cancer, has been established in both pre-clinical and human clinical trials conducted by the NCI. Lung cancer is characterized as small cell and non-small cell lung cancer (“NSCLC”). NSCLC is the most common type of lung cancer. VAL-083 has demonstrated activity against NSCLC in laboratory studies. VAL-083 was also investigated in a number of clinical trials in the United States and Europe during the 1970s both as a stand-alone therapy and in combination with other chemotherapeutic regimens. VAL-083 has been approved by the Chinese Food and Drug Administration (“CFDA”) (formerly the State Food and Drug Administration) for the treatment of lung cancer in China. However, we believe that the use of the drug in the modern era has been limited by a preference for targeted therapies.
In November 2013, we presented non-clinical data which supports the potential utility of VAL-083 in the context of modern lung cancer therapy at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics Annual Meeting. In April 2014, we presented additional data at the AACR Annual Meeting demonstrating that VAL-083 may exhibit superior efficacy against NSCLC cell lines versus standard of care in an established murine (mouse) xenograft model.
We plan to establish a strong scientific and clinical rationale to support out-licensing activities to unlock the potential value of the drug in partnership with larger pharmaceutical companies with the resources and commercial infrastructure to effectively develop and launch a lung cancer product.
Additional Orphan Drug Indications
We have established a high-level scientific rationale for the development of VAL-083 in additional high-value orphan cancer indications. Hematologic cancers such as chronic myelogenous leukemia (“CML”), acute myeloid leukemia (“AML”) are of particular interest based on published human clinical data and lack of effective therapeutic options. We have initiated preliminary discussions with leading cancer researchers regarding the development of a clinical strategy for the development of VAL-083 in hematologic cancers.
In addition to our clinical development activities in the United States, we have obtained certain exclusive commercial rights to VAL-083 in China. In October 2012, we announced that we had entered into a collaboration agreement with the only manufacturer licensed by the Chinese State Food and Drug Administration to produce the product for the China market. This agreement provides us with certain exclusive commercial rights related to drug supply, which positions us with the potential to generate near-term revenue through product sales or royalties for the drug’s approved indications in China while we seek global approval in new indications. Our strategy in China is to develop new clinical and non-clinical data in collaboration with leading cancer researchers to demonstrate the utility of VAL-083 in the treatment of CML and lung cancer, particularly for patients who do not respond to, or cannot access, modern treatments such as tyrosine kinase inhibitors. Management believes the data, if favorable, will allow the repositioning of VAL-083 in the China market, and eventually global markets, for the treatment of hematologic cancers and solid tumors. We anticipate seeking a marketing partner for VAL-083 in China in order to obtain royalty revenue from that market.
We have filed a broad portfolio of new patent applications to protect our intellectual property. Our patent applications claim compositions and methods related to the use of VAL-083 and related compounds as well as methods of synthesis and quality controls for the manufacturing process of VAL-083. In July 2013, our first patent in the United States claiming methods of synthesis for VAL-083 was issued by the United States Patent Office. We continue to prosecute patent cases in the United States and international jurisdictions.
In addition to new patent filings, we intend to seek orphan drug protection and other statutory protection for our intellectual property. In February, 2012, we announced that VAL-083 has been granted Orphan Drug protection for the treatment of glioma, including GBM by the FDA in the United States. In January 2013, the European Medicines Association (“EMA”) granted Orphan Drug protection to VAL-083. The orphan drug designation means that we may sell VAL-083 as a treatment for GBM without competition for seven years in the United States and for ten years in the European Union following market approval, in respect of a medicinal product containing a similar active substance for the same indication.
Drugs granted orphan drug protection generally follow the same regulatory development path as any other pharmaceutical product. However, incentives such as scientific advice and reduction or waiver of registration fees and access to specialized grant funding may be available to support and accelerate development of orphan drug candidates.
Developing Partnerships with Pharmaceutical Companies
Guangxi Wuzhou Pharmaceutical Company
We have a strategic collaboration with Guangxi Wuzhou Pharmaceutical Company (“Guangxi Wuzhou Pharmaceuticals”), a subsidiary of publicly traded Guangxi Wuzhou Zhongheng Group Co., Ltd for the development of VAL-083 (marketed as “DAG” in China). Guangxi Wuzhou Pharmaceuticals has received regulatory approval by the CFDA to manufacture and sell VAL-083 in China as a cancer chemotherapy for the treatment of CML and lung cancer.
We are party to a memorandum of understanding and collaboration agreement, dated October 25, 2012 (the “Guangxi Agreement”), with Guangxi Wuzhou Pharmaceuticals. Pursuant to the Guangxi Agreement, we granted to Guangxi Wuzhou Pharmaceuticals a royalty-free license to certain of our intellectual property, as it relates to quality control and drug production methods for VAL-083, and we agreed that Guangxi Wuzhou Pharmaceuticals will be our exclusive supplier of VAL-083 for clinical trials and sales for the China, United States, Canadian and European markets, subject to Guangxi Wuzhou Pharmaceuticals obtaining and maintaining cGMP certification by the FDA, EMEA or other applicable regulatory agencies, and Guangxi Wuzhou Pharmaceuticals being able to meet volumes ordered by us. Guangxi Wuzhou Pharmaceuticals agreed that it may not sell VAL-083 for markets outside of China to any other purchaser other than us. In addition, Guangxi Wuzhou Pharmaceuticals granted us a pre-emptive right (subject to our acceptance of proposed sales volume and prices) to purchase VAL-083 produced by Guangxi Wuzhou Pharmaceuticals. The collaboration under the Guangxi Agreement establishes an exclusive supply relationship between us and Guangxi Wuzhou Pharmaceuticals to include the Chinese market and all markets outside China. DelMar and Guangxi Wuzhou Pharmaceuticals will work together to ensure the product specifications meet global standards in order to accelerate international development and regulatory approval. Subject to meeting and maintaining cGMP certification, Guangxi Wuzhou Pharmaceuticals will be our exclusive supplier of DAG for injection for clinical development and commercial sales.
The Company and Guangxi Wuzhou Pharmaceuticals plan to develop new clinical data to expand the market in China and to seek regulatory approval for the drug in multiple indications on a global basis. The companies have formed a clinical advisory board to oversee clinical studies. Guangxi Wuzhou Pharmaceuticals will provide funding support for clinical trials conducted in China and we will be responsible for development and commercialization. We are currently seeking to establish a separate collaboration for the distribution, sales and marketing of VAL-083 in China.
The term of the Guangxi Agreement (except as it relates to the exclusive rights in the China market) is indefinite, subject to termination upon written agreement of all parties, or if either party breaches any material term and fails to remedy such breach within 30 days of receipt of notice of the breach, or if any action to be taken thereunder is not agreed to by both parties, provided that such matter is referred to the chief executive officer of both parties, and they are unable to resolve such matter within 90 days. No payments have been made to date under the Guangxi Agreement.
The protection of intellectual property rights in China (where VAL-083 is manufactured pursuant to the Guangxi Agreement with the only manufacturer presently licensed by the SDFA to produce the product for the China market, and where VAL-03 is approved for the treatment of CML and lung cancer) is relatively weak compared to the United States, which may negatively affect our ability to generate revenue from VAL-083.
On January 25, 2013 (the “Closing Date”), the Company entered into and closed an exchange agreement (the “Exchange Agreement”), with DelMar (BC), Callco, Exchangeco, and the securityholders of DelMar (BC). Pursuant to the Exchange Agreement, (i) the Company issued 4,340,417 shares of common stock (the “Parent Shares”) to the shareholders of DelMar (BC) who are United States residents (the “U.S. Holders”) in exchange for the transfer to Exchangeco of all 4,340,417 outstanding common shares of DelMar (BC) held by the U.S. Holders, (ii) the shareholders of DelMar (BC) who are Canadian residents (the “Canadian Holders”) received, in exchange for the transfer to Exchangeco of all 8,729,583 outstanding common shares of DelMar (BC) held by the Canadian Holders, 8,729,583 exchangeable shares (the “Exchangeable Shares”) of Exchangeco, and (iii) outstanding warrants to purchase 3,360,000 common shares of DelMar (BC) and outstanding options to purchase 1,020,000 common shares of DelMar (BC) were deemed to be amended such that, rather than entitling the holder to acquire common shares of DelMar (BC), such options and warrants will entitle the holders to acquire shares of common stock of the Company. The Canadian Holders will be entitled to require Exchangeco to redeem (or, at the option of the Company or Callco, to have the Company or Callco purchase) the Exchangeable Shares, and upon such redemption or purchase to receive an equal number of shares of common stock of the Company. The aggregate of 13,070,000 shares of common stock of the Company issued to the former shareholders of DelMar (BC) (on an as-exchanged basis with respect to the Exchangeable Shares) represented 80.1% of the outstanding shares of common stock of the Company following the closing of the Exchange Agreement (the “Reverse Acquisition”).
Upon completion of the Reverse Acquisition DelMar (BC) became a wholly-owned subsidiary of the Company. As a result of the shareholders of DelMar (BC) having a controlling interest in the Company subsequent to the Reverse Acquisition, for accounting purposes the transaction is a capital transaction with DelMar (BC) being the accounting acquirer even though the legal acquirer is Berry. No goodwill is recorded with respect to the transaction as it does not constitute a business combination. For accounting purposes, the transaction is reflected as a recapitalization of DelMar (BC) and consideration for the Reverse Acquisition was deemed to be the fair value of the shares that were issued by DelMar (BC) to acquire the net liabilities of Berry on January 25, 2013. The net identifiable liabilities of Berry on the Closing Date of the Reverse Acquisition were as follows:
|
Net liabilities (derivative liability)
|
|
|
$
|
2,041,680
|
|
The Company determined the fair value of the shares issued on the Reverse Acquisition to be $1,690,004. As a result of the Reverse Acquisition being treated as a recapitalization of DelMar (BC) the Company recognized the loss of $3,731,684 incurred upon the closing of the Reverse Acquisition as an adjustment to opening deficit in the consolidated statement of stockholder’s deficiency at December 31, 2013.
Unit Offering
In connection with the Reverse Acquisition, on January 25, 2013, January 31, 2013, February 8, 2013, February 21, 2013, February 28, 2013, March 1, 2013, and March 6, 2013, the Company entered into and closed a series of subscription agreements with accredited investors (the “Investors”), pursuant to which the Company issued an aggregate of 13,125,002 Units at a purchase price of $0.80 per Unit, for aggregate gross proceeds of $10,500,000 (the “Private Offering”). Each Unit consists of one share of common stock and one five-year warrant (the “Investor Warrants”) to purchase one share of common stock at an exercise price of $0.80. The exercise price of the Investor Warrants is subject to adjustment in the event that the Company sells common stock at a price lower than the exercise price, subject to certain exceptions. The Investor Warrants are redeemable by the Company at a price of $0.001 per Investor Warrant at any time subject to the conditions that (i) the Company’s common stock has traded for twenty (20) consecutive trading days with a closing price of at least $1.60 per share with an average trading volume of 50,000 shares per day and (ii) the underlying shares of common stock are registered.
The Company retained Charles Vista, LLC (the “Placement Agent”) as the Placement Agent for the Private Offering. The Company paid the Placement Agent a cash fee of $1,050,000 (equal to 10% of the gross proceeds), a non-accountable expense allowance of $315,000 (equal to 3% of the gross proceeds), and a one-year consulting fee of $60,000. In addition, the Company incurred other closing costs of approximately $500,000 resulting in net proceeds to the Company of $8,575,000. Certain of the additional closing costs were not eligible to be treated as share issue costs and as a result they have been expensed. Net unit proceeds per the consolidated statements of cash flows include gross unit proceeds less cash issue costs attributable to the common stock only. The portion of the unit issue costs attributable to the derivative liability has been expensed.
In addition, the Company issued to the Placement Agent five-year warrants (the “Placement Agent Warrants”) to purchase 5,250,000 shares of common stock (equal to 20% of the shares of common stock (i) included as part of the Units sold in the Private Offering and (ii) issuable upon exercise of the Investor Warrants) at an exercise price of $0.80, exercisable on a cash or cashless basis.
The Company will pay a warrant commission of 5% of the amount of funds raised by an agent upon the exercise of the Investor Warrants following such redemption.
Certain of the Private Offering costs were incurred by the Company prior to December 31, 2012. These costs of $90,771 were treated as share issue costs during the three months ended March 31, 2013.
The Company acquired its VAL-083 prototype drug, patents and technology rights from Valent. In addition, Valent has incurred a significant portion of the Company’s clinical expenses during the periods ended December 31, 2011 and 2012 and has in turn invoiced the Company for those expenses. One of the Company’s officers and directors is also a Principal of Valent and as result Valent is a related party to the Company.
The following related party transactions and balances have been recorded by the Company.
During the three months ended March 31, 2014
Pursuant to consulting agreements with the Company’s officers the Company pays a total of $32,000 per month in cash compensation to its officers. Pursuant to these agreements the Company recognized a total of $96,000 in compensation expense.
Included in accounts payable at March 31, 2014 is an aggregate amount owing of $35,798 (December 31, 2013 - $74,754) to the Company’s officers and directors for fees and expenses. The Company pays related party payables incurred for fees and expenses under normal commercial terms.
The Company paid $24,000 in directors’ fees.
During the three months ended March 31, 2013
Pursuant to consulting agreements with the Company’s officers and directors the Company pays a total of $36,784 per month to its officers and directors. Pursuant to these agreements the Company recognized a total of $110,352 in compensation expense.
On January 25, 2013, in connection with the Reverse Acquisition, Valent was issued 1,150,000 shares of common stock of the Company in exchange for Valent reducing certain future royalties under its Assignment Agreement with the Company. As a result of the share issuance the Company has recognized an expense of $598,000.
Derivative Liability
The Company has issued stock purchase warrants. Based on the terms of certain of these warrants the Company determined that the warrants are a derivative liability which is recognized at fair value at the date of the transaction and re-measured at fair value every reporting period with gains or losses on the changes in fair value recorded in the consolidated condensed interim statement of loss and comprehensive loss.
CDN $0.50 Unit Warrants
The Company issued 4,150,000 units on January 23, 2012, 560,000 on February 27, 2012 and 50,000 on May 10, 2012. In addition, during the year ended December 31, 2011 the Company issued 500,000 units on October 3, 2011, 100,000 on October 7, 2011, and 50,000 on November 11, 2011. In total, the Company issued 5,410,000 units for services in settlement of accounts payable and cash proceeds for an aggregate of $2,671,923 (CDN $2,705,000).
The proceeds from the issuance of 3,000,000 of these units were held in escrow pursuant to an exclusive option investment agreement with a strategic investor. Subsequently, the Company elected to let the option expire and the related units were cancelled and the funds returned from escrow to the subscriber in order for the Company to retain control over certain intellectual property and commercial rights.
During the three months ended March 31, 2014, 20,000 warrants were exercised for no additional consideration for 20,000 shares of common stock. As a result, $17,600 of the derivative liability has been reclassified to equity. The warrants that have been exercised were revalued at their exercise date and then the reclassification to equity was recorded.
The remaining 2,169,000 warrants expired on January 25, 2014. As of March 31, 2014 there are no CDN $0.50 unit warrants outstanding.
Investor Warrants
As discussed above, in connection with the Reverse Acquisition, , the Company entered into and closed a series of subscription agreements with accredited investors (the “Investors”), pursuant to which the Company issued an aggregate of 13,125,002 Units at a purchase price of $0.80 per Unit, for aggregate gross proceeds of $10,500,000 (the “Private Offering”). Each Unit consists of one share of common stock and one five-year warrant (the “Investor Warrants”) to purchase one share of common stock at an exercise price of $0.80
During the three months ended March 31, 2014, 277,313 warrants were exercised for 277,313 shares of common stock. The Company received proceeds of $221,850 from the exercises. As a result, $126,064 of the derivative liability has been reclassified to equity. The warrants that have been exercised were revalued at their exercise date and then the reclassification to equity was recorded.
Dividend Warrants
As a result of the Reverse Acquisition warrants that Berry issued pursuant to a warrant dividend became warrants of the Company (the “Dividend Warrants”). The Dividend Warrants are exercisable at $1.25 per share until January 24, 2018. The Dividend Warrants are exercisable only at such times as the underlying shares of common stock are registered. The Dividend Warrants will be redeemable by the Company at a price of $0.001 per Dividend Warrant at any time commencing 18 months following the date of issuance subject to the conditions that (i) the Company’s common stock has traded for twenty (20) consecutive trading days with a closing price of at least $2.50 per share and (ii) the underlying shares of common stock are registered. Subject to the conditions set forth therein, the Dividend Warrants may be redeemed by the Company upon not less than sixty (60) days nor more than ninety (90) days prior written notice.
Warrants issued for services
The Company has issued 300,000 warrants for services. The warrants were issued on September 12, 2013 and are exercisable on a cashless basis at an exercise price of $1.76 for five years.
The Company’s derivative liability is summarized as follows:
| |
|
March 31,
2014
$
|
|
|
December 31,
2013
$
|
|
| |
|
|
|
|
|
|
|
Opening balance
|
|
|
4,402,306
|
|
|
|
121,000
|
|
| |
|
|
|
|
|
|
|
|
|
Issuance of units
|
|
|
-
|
|
|
|
3,681,372
|
|
|
Dividend Warrant liability acquired on reverse acquisition
|
|
|
-
|
|
|
|
2,041,680
|
|
|
Warrants issued for services
|
|
|
-
|
|
|
|
124,020
|
|
|
Change in fair value of unexercised warrants
|
|
|
1,599,349
|
|
|
|
(1,324,051
|
)
|
|
Reclassification to equity upon exercise of warrants
|
|
|
(143,664
|
)
|
|
|
(241,715
|
)
|
| |
|
|
|
|
|
|
|
|
|
Closing balance
|
|
|
5,857,991
|
|
|
|
4,402,306
|
|
Selected Quarterly Information
The financial information reported here in has been prepared in accordance with US GAAP. The Company’s functional currency at March 31, 2014 is the USD. The following table represents selected financial information for the Company as of March 31, 2014 and December 31, 2013.
Selected Balance Sheet Data
| |
|
March 31,
2014
$
|
|
|
December 31,
2013
$
|
|
| |
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
3,474,150
|
|
|
|
4,136,803
|
|
|
Working capital
|
|
|
3,313,942
|
|
|
|
4,069,261
|
|
|
Total Assets
|
|
|
3,754,153
|
|
|
|
4,318,748
|
|
|
Derivative liability
|
|
|
5,857,991
|
|
|
|
4,402,306
|
|
|
Total shareholder’s deficiency
|
|
|
(3,060,299
|
)
|
|
|
(817,978
|
)
|
Comparison of the three months ended March 31, 2014 and 2013
The following table represents selected financial information for the Company for the three months ended March 31, 2014 and March 31, 2013.
| |
|
Three Months Ended
|
|
|
|
| |
|
March 31,
2014
$
|
|
|
March 31
2013
$
|
|
Change
$
|
Change
%
|
| |
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
618,869
|
|
|
|
631,947
|
|
(13,078)
|
(2)
|
|
General and administrative
|
|
|
966,923
|
|
|
|
920,377
|
|
46,546
|
5
|
|
Change in fair value of derivative liability
|
|
|
1,599,349
|
|
|
|
2,543,574
|
|
(944,225)
|
(37)
|
|
Shares issued to Valent for future royalty reduction
|
|
|
-
|
|
|
|
598,000
|
|
(598,000)
|
(100)
|
|
Derivative issue costs
|
|
|
-
|
|
|
|
2,713,220
|
|
(2,713,220)
|
(100
|
|
Foreign exchange loss (gain)
|
|
|
11,947
|
|
|
|
(3,754)
|
|
15,701
|
(418)
|
|
Interest expense
|
|
|
2,015
|
|
|
|
1,955
|
|
60
|
3
|
|
Interest income
|
|
|
(496)
|
|
|
|
-
|
|
(496)
|
(100)
|
|
Net loss
|
|
|
3,198,607
|
|
|
|
7,405,319
|
|
(4,206,712)
|
|
| |
|
Three Months Ended
|
|
|
|
|
|
|
|
| |
|
March 31,
2014
$
|
|
|
March 31,
2013
$
|
|
|
Change
$
|
|
|
Change
%
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
618,869
|
|
|
|
631,947
|
|
|
|
(13,078
|
)
|
|
|
(2
|
)
|
|
Share-based compensation included in research and development
|
|
|
(171,947
|
)
|
|
|
(152,480
|
)
|
|
|
(19,467
|
)
|
|
|
13
|
|
|
Research and development net of share-based compensation
|
|
|
446,922
|
|
|
|
479,467
|
|
|
|
(32,545
|
)
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
General and administrative
|
|
|
966,923
|
|
|
|
920,377
|
|
|
|
46,546
|
|
|
|
5
|
|
|
Share-based compensation included in general and administrative
|
|
|
(448,127
|
)
|
|
|
(120,422
|
)
|
|
|
(327,705
|
)
|
|
|
(272
|
)
|
|
General and administrative net of share-based compensation
|
|
|
518,796
|
|
|
|
799,955
|
|
|
|
(281,159
|
)
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and Development
Research and development expenses decreased to $618,869 for the three months ended March 31, 2014 from $631,947 for the three months ended March 31, 2013. The increase was partially attributable to an increase in share-based payments to $171,947 for the three months ended March 31, 2014 compared to $152,480 for the three months ended March 31, 2013. In relation to research and development during the three months ended March 31, 2014 and 2013 the Company recognized share-based payment expense relating to the measurement of stock options granted in prior periods. The increase relating to stock option expense was due to more stock options being outstanding at March 31, 2014 compared to March 31, 2013. The effect of there being more stock options outstanding during the current period than the prior period was partially offset by a decrease in the Company’s share price during the three months ended March 31, 2014 compared to the three months ended March 31, 2013.
After considering the impact of share-based payments, research and development expenses decreased in the three months ended March 31, 2014 to $446,922 from $479,467 for the three months ended March 31, 2013. The largest portion of the non-share-based payment component of research and development for the quarter ended March 31, 2014 was clinical development costs as the Company continued its Phase I/II clinical trial with VAL-083. The clinical development costs were lower in the current quarter compared to the prior quarter largely due to the timing of patient enrollment. Intellectual property costs have decreased in the three months ended March 31, 2014 compared to the three months ended March 31, 2013 due to the timing of the filing and advancement of patent matters. In addition, travel costs decreased during the three months ended March 31, 2014 compared to the three months ended March 31, 2013 due to fewer trips for senior science staff. In the prior quarter, senior science staff travelled extensively to make scientific presentations in support of the Company’s efforts to complete its Reverse Acquisition financing. Partially offsetting the impact of lower clinical, intellectual property, and travel costs were higher pre-clinical costs in the current quarter compared to the prior quarter. Preclinical expenses were higher in the three months ended March 31, 2014 compared to the three months ended March 31, 2013 as the Company has begun to incur costs associated with advancing its NSCLC program as well as continuing its mechanism of action elucidation for VAL-083.
General and Administrative
General and administrative expenses were $966,923 for the three months ended March 31, 2014 compared to $920,377 for the three months ended March 31, 2013. The increase was largely attributable to an increase in share-based payments to $448,127 in the three months ended March 31, 2014 compared to $120,422 for the three months ended March 31, 2013. In relation to general and administrative expenses during the three months ended March 31, 2014 and 2013 the Company incurred share-based payments relating to the measurement of stock options granted in prior periods and for shares issued for services. For the three months ended March 31, 2014 the expense recognized for shares issued for services was higher than for the three months ended March 31, 2013. In addition, the increase relating to stock option expense was due to more stock options being outstanding at March 31, 2014 compared to March 31, 2013. The effect of there being more stock options outstanding during the current period than the prior period was partially offset by a decrease in the Company’s share price during the three months ended March 31, 2014 compared to the three months ended March 31, 2013.
After considering the impact of share-based payments, general and administrative expenses decreased in the three months ended March 31, 2014 to $518,796 from $799,955 for the three months ended March 31, 2013. The principal reason for the decrease was due to professional fees relating to legal, accounting, investor relations, and business development. As a result of the Company becoming public due to its Reverse Acquisition during the three months ended March 31, 2013, the Company incurred significant one-time professional fees that were not incurred during the current quarter. Partially offsetting the lower professional fees in the current quarter were higher travel, and office and sundry expenses. Travel increased during the three months ended March 31, 2014 compared to the three months ended March 31, 2013 due to the attendance at more conferences and investor meetings in 2014 compared to 2013. Office and sundry increased for the three months ended March 31, 2014 compared to the three months ended March 31, 2013 largely due an increase in insurance, and filing and related fees.
Change in fair value of derivative liability
Based on the terms of certain warrants issued by the Company, the Company determined that the warrants were a derivative liability which is recognized at fair value at the date of the transaction and re-measured at fair value every reporting period with gains or losses on the changes in fair value recorded in the consolidated condensed interim statement of loss and comprehensive loss.
The Company recognized a loss of $1,599,349 from the change in fair value of the derivative liability for the three months ended March 31, 2014 compared to a loss of $2,543,574 for the three months ended March 31, 2013. The balances recognized during the three months ended March 31, 2014 and March 31, 2013 depend on a number of factors and assumptions including volatility assumptions and the Company’s common stock price between the date the warrants were last valued and respective reporting dates.
Changes in the Company’s common stock price can result in significant volatility in the Company’s reported net loss due to its impact on the fair value of the derivative liability. As a result of revaluation gains and losses, it is expected that the Company’s reported net income or loss will continue to experience large fluctuations.
Issuance of Shares to Valent for future royalty reduction
On January 25, 2013, in connection with the Reverse Acquisition, the Company issued to Valent 1,150,000 shares of common stock in exchange for Valent agreeing to the reduction of certain future royalties payable to Valent under the Assignment Agreement. As a result of the share issuance the Company has recognized an expense of $598,000 for the three months ended March 31, 2013.
Derivative issue costs
The proceeds from the $0.80 unit offering have been allocated between common stock and derivative liability based on the respective fair values of the shares of common stock and the warrants on the issuance date. Additionally, the unit issue costs have also been allocated between common stock and derivative liability on the same pro rata basis as the proceeds. The portion of the issue costs allocated to the derivative liability has been expensed in the consolidated statement of loss and comprehensive loss. The Company recognized $2,713,220 in derivative issue costs for the three months ended March 31, 2013.
Foreign Exchange Gain
The Company’s functional currency at March 31, 2014 is the USD but the Company incurs a portion of its expenses in CDN. The foreign exchange gains and losses are reported in other (income) loss in the consolidated condensed interim statement of loss and comprehensive loss.
The Company recognized a foreign exchange loss of $11,947 for the quarter ended March 31, 2014 compared to a gain of $3,754 for the quarter ended March 31, 2013. The change was due to changes in the exchange rate between the CDN and the USD and to varying levels of CDN accounts payable.
Pursuant to a loan agreement dated February 3, 2011, the Company has received a loan from Valent in the amount of $250,000 for the purchase of the prototype drug product. The loan is payable on demand, unsecured and bears interest at 3.00% per year. As a result of the loan payable the Company recognized $2,015 and $1,955 respectively in accrued interest for the three months ended March 31, 2014 and 2013.
Liquidity and Capital Resources
Three months ended March 31, 2014 compared to the three months ended March 31, 2013
| |
|
March 31,
2014
$
|
|
|
March 31,
2013
$
|
|
|
Change
$
|
|
|
Change
%
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash used in operating activities
|
|
|
(884,503
|
)
|
|
|
(2,124,467
|
)
|
|
|
1,239,964
|
|
|
|
(58
|
)
|
|
Cash flows from financing activities
|
|
|
221,850
|
|
|
|
9,639,520
|
|
|
|
(9,417,670
|
)
|
|
|
(98
|
)
|
Comparison of cash flow for the three months ended March 31, 2014 compared to the three months ended March 31, 2013
Net cash used in operating activities decreased to $884,503 for the three months ended March 31, 2014 from $2,124,467 for the three months ended March 31, 2013. The decrease was largely the result of a decrease in the net loss to $3,198,607 for the three months ended March 31, 2014 compared to $7,405,319 for the three months ended March 31, 2013. Partially offsetting the impact of the lower net loss were non-cash items totaling $2,221,438 incurred in the current period consisting of accrued loan interest, change in the fair value of the derivative liability and share-based payments. The non-cash items for the three months ended March 31, 2013 were significantly higher and totaled $5,617,439 and consisted of accrued loan interest, change in the fair value of the derivative liability, expense related to shares issued to Valent for the future royalty reduction, non-cash derivative issue costs, and share-based payments. The most significant changes in non-cash working capital for the three months ended March 31, 2014 were an inflow from the increase in accounts payable and accrued liabilities of $263,956 and an outflow of $98,701 for the payment of prepaid expenses. In the three months ended March 31, 2013 there were outflows of $151,718 and $98,722 from increases in accounts payable and accrued liabilities, and related party payables respectively.
As a result of the Company’s expectations as to the timing of the repayment of the Valent loan, the Company has presented the full loan and accrued interest balance as a long-term liability at March 31, 2014 and December 31, 2013.
The only financing activities for the three months ended March 31, 2014 was the receipt of $221,850 in proceeds from the exercise of 277,313 $0.80 Investor Warrants. During the three months ended March 31, 2013 the Company received $9,639,520 in net proceeds from the issuance of units. The net cash proceeds from the unit financing were $8,575,000. However, as a result of a portion of the unit proceeds and issue costs being accounted for as a derivative liability the net proceeds on the condensed consolidated statement of cash flows is $9,639,520. During the three months ended March 31, 2013 certain of the additional closing costs were not eligible to be treated as share issue costs and as a result they have been expensed. Net unit proceeds per the condensed consolidated interim statements of cash flows include gross unit proceeds less cash issue costs attributable to the shares only. The portion of the unit issue costs attributable to the derivative liability has been expensed.
The units issued in the three months ended March 31, 2013 were the $0.80 units issued in conjunction with the Reverse Acquisition.
Operating Capital and Capital Expenditure Requirements
Liquidity and capital resources (also see ‘Going Concern’ section)
Our future funding requirements will depend on many factors, including but not limited to:
|
·
|
the rate of progress and cost of our clinical trials, preclinical studies and other discovery and research and development activities;
|
|
·
|
the costs associated with establishing manufacturing and commercialization capabilities;
|
|
·
|
the costs of acquiring or investing in businesses, product candidates and technologies;
|
|
·
|
the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights;
|
|
·
|
the costs and timing of seeking and obtaining FDA and other regulatory approvals;
|
|
·
|
the effect of competing technological and market developments; and
|
|
·
|
the economic and other terms and timing of any collaboration, licensing or other arrangements into which we may enter.
|
Until we can generate a sufficient amount of product revenue to finance our cash requirements, which we may never do, we expect to finance future cash needs primarily through public or private equity offerings, debt financings or strategic collaborations. Although we are not reliant on institutional credit finance and therefore not subject to debt covenant compliance requirements or potential withdrawal of credit by banks, the current economic climate has also impacted the availability of funds and activity in equity markets. We do not know whether additional funding will be available on acceptable terms, or at all. If we are not able to secure additional funding when needed, we may have to delay, reduce the scope of or eliminate one or more of our clinical trials or research and development programs or make changes to our operating plan. In addition, we may have to seek partnerships for one or more of our product candidate programs at an earlier stage of development, which would lower the economic value of those programs to us.
Critical Accounting Policies
The preparation of financial statements, in conformity with generally accepted accounting principles in the United States, requires companies to establish accounting policies and to make estimates that affect both the amount and timing of the recording of assets, liabilities, revenues and expenses. Some of these estimates require judgments about matters that are inherently uncertain and therefore actual results may differ from those estimates.
|
A detailed summary of all of the Company’s significant accountings policies and the estimates derived therefrom is included in Note 2 to the Company’s Financial Statements for the year ended December 31 2013. While all of the significant accounting policies are important to the Company’s consolidated financial statements, the following accounting policies and the estimates derived therefrom have been identified as being critical:
|
| |
|
· Shares for services
|
|
· Stock options
|
|
· Derivative liability
|
The Company has issued equity instruments for services provided by employees and nonemployees. The equity instruments are valued at the fair value of the instrument granted (see notes 6 and 7 of the consolidated financial statements for assumptions).
In prior periods the Company transferred shares from the DelMar Employee Share Purchase Trust (the “Trust”) to consultants and management in exchange for services rendered to the Company. The Company recognizes the fair value of the shares transferred as an expense with a corresponding increase in common stock. The shares reserved for issuance to consultants and management that are held by the Trust are included in the financial statements at year end. There are no other assets in the Trust. The number of shares outstanding for issue from the Trust at December 31, 2013 is nil (December 31, 2012 – nil).
The shares transferred from the Trust in prior periods have been valued using the fair value of the shares transferred. The Company has used recent share transactions in order to determine the fair value of the shares transferred from the Trust.
The Company accounts for these awards under ASC 718, “Compensation - Stock Compensation” (“ASC 718”). ASC 718 requires measurement of compensation cost for all stock-based awards at fair value on the date of grant and recognition of compensation over the requisite service period for awards expected to vest. Compensation expense for unvested options to non-employees is revalued at each period end and is being amortized over the vesting period of the options. The determination of grant-date fair value for stock option awards is estimated using the Black-Scholes model, which includes variables such as the expected volatility of the Company’s share price, the anticipated exercise behavior of its grantee, interest rates, and dividend yields. These variables are projected based on the Company’s historical data, experience, and other factors. Changes in any of these variables could result in material adjustments to the expense recognized for share-based payments. Such value is recognized as expense over the requisite service period, net of estimated forfeitures, using the straight-line attribution method. The estimation of stock awards that will ultimately vest requires judgment, and to the extent actual results or updated estimates differ from current estimates, such amounts are recorded as a cumulative adjustment in the period estimates are revised. The Company considers many factors when estimating expected forfeitures, including type of awards granted, employee class, and historical experience. Actual results and future estimates may differ substantially from current estimates.
The Company accounts for certain warrants under the authoritative guidance on accounting for derivative financial instruments indexed to, and potentially settled in, a company’s own stock, on the understanding that in compliance with applicable securities laws, the warrants require the issuance of securities upon exercise and do not sufficiently preclude an implied right to net cash settlement. The Company classifies warrants in its balance sheet as a derivative liability which is fair valued at each reporting period subsequent to the initial issuance. As quoted prices for the derivative liability are not available, the Company uses a simulated probability valuation model to value the warrants. Determining the appropriate fair-value model and calculating the fair value of warrants requires considerable judgment. Any change in the estimates used may cause the value to be higher or lower than that reported. The estimated volatility of the Company’s common stock at the date of issuance, and at each subsequent reporting period, is based on the historical volatility of similar life sciences companies. The risk-free interest rate is based on rates published by the government for bonds with a maturity similar to the expected remaining life of the warrants at the valuation date. The expected life of the warrants is assumed to be equivalent to their remaining contractual term.
Off-Balance Sheet Arrangements
We do not have any off balance sheet arrangements.
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
DIRECTORS AND EXECUTIVE OFFICERS
Below are the names and certain information regarding the Company’s executive officers and directors.
|
Name
|
|
|
Age
|
|
Position
|
| |
|
|
|
|
|
|
Jeffrey Bacha
|
|
|
46
|
|
President, Chief Executive Officer and Director
|
| |
|
|
|
|
|
|
Dennis Brown
|
|
|
64
|
|
Chief Scientific Officer and Director
|
| |
|
|
|
|
|
|
Scott Praill
|
|
|
47
|
|
Chief Financial Officer
|
| |
|
|
|
|
|
|
William Garner
|
|
|
47
|
|
Director
|
| |
|
|
|
|
|
|
John K. Bell
|
|
|
66
|
|
Director
|
| |
|
|
|
|
|
|
Robert J. Toth, Jr.
|
|
|
50
|
|
Director
|
Jeffrey Bacha, BSc, MBA has been Chief Executive Officer and President of the Company since January 25, 2013, and director of the Company since February 11, 2013. Mr. Bacha is one of our founders and has been President, Chief Executive Officer and director of DelMar (BC) since inception. Mr. Bacha is a seasoned executive leader with nearly twenty years of life sciences experience in the areas of operations, strategy and finance. His background includes successful public and private company building from both a start-up and turn around perspective; establishing and leading thriving management and technical teams; and raising capital in both the public and private markets. From July 2006 to August 2009, Mr. Bacha was Executive Vice President Corporate Affairs and Chief Operating Officer at Clera, Inc. From March 2005 to July 2006 Mr. Bacha was Consultant and held various positions at Clera Inc., Urigen Holdings Inc. and XBiotech, Inc. From 1999 through 2004, Mr. Bacha served as President & CEO of Inimex Pharmaceuticals, a venture-capital funded drug discovery and development company and is a former Senior Manager and Director of KPMG Health Ventures. Mr. Bacha holds an MBA from the Goizueta Business School at Emory University and a degree in BioPhysics from the University of California, San Diego. Mr. Bacha’s experience as one of our founder and Chief Executive Officer qualifies him to serve on the Board of Directors.
Dr. Dennis M. Brown, PhD, has been Chief Scientific Officer of the Company since January 25, 2013 and director of the Company since February 11, 2103. Dr. Brown is one of our founders and has served as Chief Scientific Officer and director of DelMar (BC) since inception. Dr. Brown has more than thirty years of drug discovery and development experience. He has served as Chairman of Mountain View Pharmaceutical's Board of Directors since 2000 and is the President of Valent. In 1999 he founded ChemGenex Therapeutics, which merged with a publicly traded Australian company in 2004 to become ChemGenex Pharmaceuticals (ASX: CXS/NASDAQ: CXSP), of which he served as President and a Director until 2009. He was previously a co-founder of Matrix Pharmaceutical, Inc., where he served as Vice President (VP) of Scientific Affairs from 1985-1995 and as VP, Discovery Research, from 1995-1999. He also previously served as an Assistant Professor of Radiology at Harvard University Medical School and as a Research Associate in Radiology at Stanford University Medical School. He received his B.A. in Biology and Chemistry (1971), M.S. in Cell Biology (1975) and Ph.D. in Radiation and Cancer Biology (1979), all from New York University. Dr. Brown is an inventor of about 34 issued U.S. patents and applications, many with foreign counterparts. Dr. Brown’s scientific knowledge and experience qualifies him to serve on our Board of Directors.
Scott Praill, CPA, BSc. has been Chief Financial Officer of the Company since January 29, 2013 and previously served as a consultant to DelMar (BC). Since 2004, Mr. Praill has been an independent consultant providing accounting and administrative services to companies in the resource industry. Mr. Praill served as CFO of Strata Oil & Gas, Inc. from June 2007 to September 2008. From November 1999 to October 2003 Mr. Praill was Director of Finance at Inflazyme Pharmaceuticals Inc. Mr. Praill completed his articling at Price Waterhouse (now PricewaterhouseCoopers LLP) and obtained his Chartered Accountant designation in 1996. Mr. Praill obtained his Certified Public Accountant (Illinois) designation in 2001. Mr. Praill received a Financial Management Diploma (Honors), from British Columbia Institute of Technology in 1993, and a Bachelor of Science from Simon Fraser University in 1989.
Dr. William Garner, MD, MPH has served as a director of the Company since February 11, 2013. Dr. Garner is one of our founders and has served as a director of DelMar (BC) since inception. Dr. Garner is an experienced entrepreneur and investor. He is founder and managing director of EGB Advisors, LLC ("EGB"), a pharmaceutical commercialization boutique. Through this entity, Dr. Garner has worked on a number of pharmaceutical business transactions and has raised financing for several drug development companies including Update Pharma, Inc. where he is currently Executive Chairman. Other EGB companies include Urigen Pharmaceuticals, Inc., and Inverseon, Inc., which is developing a novel therapy for smoking cessation, asthma and other pulmonary diseases. In 2012, he merged Inverseon with another company to form Invion Ltd. (ASX:IVX), serving as CEO until May of 2013. He also served as President and Chief Executive Officer of Urigen Pharmaceuticals, Inc. (URGP.PK) from December 2005 to December 2010 where he moved a procedure-based drug from a university license to a phase II multi-center clinical trial which achieved statistical significance on all end points in Painful Bladder Syndrome/Interstitial Cystitis. Before this, Dr. Garner worked in medical affairs at Hoffmann LaRoche in oncology. Prior to Roche, Dr. Garner was in the venture capital department at Paramount Capital Investments in New York City. He serves on the boards of ImmunoGenetix in Kansas City and the Innovation Angel Foundation in San Francisco. Dr. Garner has a Master of Public Health from Harvard and received his M.D. degree from New York Medical College. Dr. Garner did residency training in Anatomic Pathology at Columbia-Presbyterian and is currently a licensed physician in the State of New York. Dr. Garner’s medical and scientific knowledge and experience qualifies him to serve on our board of directors.
John K. Bell, FCPA, FCA, ICD.D has served as a director of the Company since February 11, 2013. John K. Bell is Chairman of Onbelay Capital Inc, a Canadian based private equity company with principal investments in Telematics and auto parts manufacturing (for past 5 years). Prior to that, from 1996 to 2005, Mr. Bell was CEO and owner of Polymer Technologies Inc., an automotive parts manufacturer. Prior to that, from 1977 to 1995, Mr. Bell was founder and owner of Shred-Tech Limited a global manufacturer and supplier of industrial shredders and mobile document shredders. Mr. Bell served as interim CEO and director of ATS Automation Tooling Systems (TSX-ATA) in 2007. Mr. Bell is a director of BSM Wireless (TSX-GPS), Strongco Corporation (TSX-SQP), and the Royal Canadian Mint (TSX-MNT). Mr. Bell is National secretary and board member of The Crohns and Colitis Foundation of Canada. Mr. Bell is past Chairman of Waterloo Regional Police, Cambridge Memorial Hospital, Canada’s Technology Triangle accelerator network and The Region of Waterloo prosperity counsel. Mr. Bell is a graduate of Western University Ivey School of Business, a Fellow of the institute of Chartered Accountants of Ontario, a graduate of the Institute of Directors Program of Canada and the owner’s president program at Harvard and International marketing program at Oxford. Mr. Bell’s financial and executive business experience qualifies him to serve on our board of directors.
Robert J. Toth, Jr., MBA has served as a director of the Company since August 2013. Since 2005, Mr. Toth has primarily been managing his personal investment portfolio. From 2004-2005, Mr. Toth served as a consulting analyst to Narragansett Asset Management, a New York-based healthcare-focused hedge fund, where he advised the firm on biotechnology investments. From 2001-2003, he was the Senior Portfolio Manager for San Francisco-based EGM Capital’s Medical Technology hedge fund, where he was responsible for managing and maintaining a dedicated medical technology portfolio. Mr. Toth began his Wall Street career in 1996 as an Equity Research Associate for Vector Securities International, a healthcare-focused brokerage firm. From 1997–1999 he served as Senior Biotechnology Analyst. He joined Prudential Securities as Senior Vice President and Biotechnology Analyst where he served from 1999-2001 following Prudential’s acquisition of Vector. His responsibilities included the analysis of commercial, clinical and scientific fundamentals of oncology- and genomics-based biotechnology companies on behalf of institutional investors. Mr. Toth was named to the Wall Street Journal’s Allstar List for stock picking in 1999. Mr. Toth received an MBA from the University of Washington and Bachelor of Science degrees in Biological Sciences and Biochemistry from California Polytechnic State University, San Luis Obispo. Mr. Toth’s financial and biotechnology industry knowledge and experience quality him to serve on the Company’s board of directors.
The Company’s directors are elected at the annual meeting of shareholders to hold office until the annual meeting of shareholders for the ensuing year or until their successors have been duly elected and qualified. Officers are elected annually by the Board of Directors and serve at the discretion of the Board.
The Company’s executive officers are not full-time employees, but are engaged by us on an independent contractor or contract-employment basis. Mr. Bacha and Mr. Praill each devote 100% of their business time to us, and Dr. Brown devotes approximately 80% of his business time to us. See “Executive Compensation”.
Board Leadership Structure and Role in Risk Oversight
Due to the small size and early stage of the Company, we have not adopted a formal policy on whether the Chairman and Chief Executive Officer positions should be separate or combined. Since Lisa Guise's resignation, these roles have been combined with Mr. Bacha serving as Chief Executive Officer and Chairman.
Our Board of Directors is primarily responsible for overseeing our risk management processes on behalf of the Company. The Board of Directors receives and reviews periodic reports from management, auditors, legal counsel, and others, as considered appropriate regarding the Company’s assessment of risks. The Board of Directors focuses on the most significant risks facing our company and our company’s general risk management strategy, and also ensures that risks undertaken by the Company are consistent with the board’s appetite for risk. While the board oversees the Company’s risk management, management is responsible for day-to-day risk management processes. We believe this division of responsibilities is the most effective approach for addressing the risks facing our company and that our board leadership structure supports this approach.
Involvement in Certain Legal Proceedings
To our knowledge, our directors and executive officers have not been involved in any of the following events during the past ten years:
| |
1.
|
any bankruptcy petition filed by or against such person or any business of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time;
|
| |
|
|
| |
2.
|
any conviction in a criminal proceeding or being subject to a pending criminal proceeding (excluding traffic violations and other minor offenses);
|
| |
|
|
| |
3.
|
being subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining him from or otherwise limiting his involvement in any type of business, securities or banking activities or to be associated with any person practicing in banking or securities activities;
|
| |
|
|
| |
4.
|
being found by a court of competent jurisdiction in a civil action, the SEC or the Commodity Futures Trading Commission to have violated a Federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated;
|
| |
|
|
| |
5.
|
being subject of, or a party to, any Federal or state judicial or administrative order, judgment decree, or finding, not subsequently reversed, suspended or vacated, relating to an alleged violation of any Federal or state securities or commodities law or regulation, any law or regulation respecting financial institutions or insurance companies, or any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or
|
| |
|
|
| |
6.
|
being subject of or party to any sanction or order, not subsequently reversed, suspended, or vacated, of any self-regulatory organization, any registered entity or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member.
|
The Board has formed an audit committee, which currently consists of John K. Bell, Chair, and Rob Toth. The Board intends to expand the audit committee at such time as the Board has additional independent members. Currently, the Board does not have any standing nominating or compensation committees, or committees performing similar functions. The functions customarily performed by such committees have been performed by the Company’s Board of Directors.
Board Leadership Structure and Role in Risk Oversight
We have not adopted a formal policy on whether the Chairman and Chief Executive Officer positions should be separate or combined. These roles are currently combined, with Mr. Bacha serving as Chief Executive Officer and Chairman.
Our board of directors is primarily responsible for overseeing our risk management processes. The board of directors receives and reviews periodic reports from management, auditors, legal counsel, and others, as considered appropriate regarding our company’s assessment of risks. The board of directors focuses on the most significant risks facing our company and our company’s general risk management strategy, and also ensures that risks undertaken by our Company are consistent with the board’s appetite for risk. While the board oversees our company’s risk management, management is responsible for day-to-day risk management processes. We believe this division of responsibilities is the most effective approach for addressing the risks facing our company and that our board leadership structure supports this approach.
During its last two fiscal years, Berry did not pay any compensation to its officers or directors.
The following table sets forth all compensation paid in respect of the Company’s principal executive officers and those individuals who received compensation in excess of $100,000 per year for 2013 and 2012. No other officer of the Company received compensation in excess of $100,000 for 2013 and 2012.
|
Name and Principal Position
|
|
Year
|
|
Salary (US$)
|
|
|
Option Awards (US$)
|
|
|
Total (US$)
|
|
|
Jeffrey Bacha CEO
|
|
2013
|
|
|
139,871
|
|
|
|
199,850
|
(2)
|
|
|
339,721
|
|
| |
|
2012
|
|
|
144,072
|
|
|
|
45,832
|
(1)
|
|
|
189,904
|
|
|
Dennis Brown Chief Scientific Officer
|
|
2013
|
|
|
120,000
|
|
|
|
199,850
|
(2)
|
|
|
319,850
|
|
| |
|
2012
|
|
|
120,000
|
|
|
|
45,832
|
(1)
|
|
|
165,832
|
|
|
Scott Praill, Chief Financial Officer
|
|
2013
|
|
|
136,399
|
|
|
|
199,850
|
(3)
|
|
|
336,249
|
|
| |
(1) Represents the grant date fair value of 150,000 options with an exercise price of Cdn $0.50 issued on February 1, 2012. The options vested over a 12 month period and expire 10 years from the date of grant. Please see Note 8 to the financial statements.
(2) Represents the grant date fair value of 350,000 options with an exercise price of $1.05 issued on August 15, 2013. The options vested over a 12 month period and expire 10 years from the date of grant. Please see Note 8 to the financial statements.
(3) Represents the grant date fair value of 350,000 options with an exercise price of $1.05 issued on August 15, 2013. The options vested over a 36 month period and expire 10 years from the date of grant. Please see Note 8 to the financial statements.
|
Pursuant to consulting agreements dated August 1, 2011 with certain of DelMar (BC)’s officers and directors at that time, DelMar (BC) agreed to compensate its officers and directors for services rendered to it, in the amount of an aggregate of Cdn $27,000 ($12,000 for Mr. Bacha, $10,000 for Dr. Brown, and $5,000 for Dr. Garner) per month commencing August 1, 2011 and ending December 31, 2012. Under the consulting agreements, DelMar (BC) and the respective officer or director mutually agreed that a portion of the compensation payable under the respective agreement for the year ended December 31, 2011 shall be deemed to have been invested in the unit offering of DelMar (BC) completed on October 3, 2011.
The consulting agreements between DelMar (BC) and each of the three executive officers and directors expired on December 31, 2012. We have continued to compensate Mr. Bacha, Dr. Brown, and Dr. Garner to December 31, 2013 at the rates set forth in their respective consulting agreements, and Mr. Bacha, Dr. Brown and Dr. Garner have continued to provide services to us as Chief Executive Officer, Chief Scientific Officer, and director, respectively. Mr. Bacha devotes 100% of his business time to us and Dr. Brown devotes approximately 80% of his business time to us. The expired consulting agreements between DelMar (BC) and Mr. Bacha and Dr. Brown, respectively, did not specify the amount of time Mr. Bacha and Dr. Brown were required to devote to us, but did require that Mr. Bacha and Dr. Brown each provide us with the full benefit of their respective knowledge, expertise and ingenuity, and prohibited Mr. Bacha and Dr. Brown from engaging in any business, enterprise or activity contrary to or that would detract from our business.
Under two of these agreements for the year ended December 31, 2012, the directors elected to receive a portion of their aggregate compensation in the form of units. During the year ended December 31, 2012 DelMar (BC) issued 360,000 units for a total amount of Cdn $180,000. The units issued relate to an amount of Cdn $15,000 per month from January to December 2012 inclusive.
The Company anticipates entering into employment agreement with Mr. Bacha and Dr. Brown in the near future.
We entered into a consulting agreement, dated February 1, 2013, with Scott Praill, our Chief Financial Officer. Pursuant to the consulting agreement, we agreed to pay Mr. Praill a fee of Cdn $10,000 per month and a one-time start-up fee of Cdn $30,000 for services rendered to date. The consulting agreement did not specify the amount of time Mr. Praill is required to devote to us, but did require that Mr. Praill provide us with the full benefit of his knowledge, expertise and ingenuity, and prohibited Mr. Praill from engaging in any business, enterprise or activity contrary to or that would detract from our business. The consulting agreement expired on December 31, 2013. Mr. Praill devotes 100% of his business time to us. To date, the consulting agreement has not been renewed but we continue to compensate Mr. Praill under the terms of the original agreement and Mr. Praill continues to serve as our Chief Financial Officer.
Outstanding Equity Awards at Fiscal Year-End
Berry had no outstanding equity awards or equity compensation plan as of December 31, 2012. Effective as of the closing of the Reverse Acquisition on January 25, 2013, outstanding options to purchase 1,020,000 common shares of DelMar (BC) were deemed to be amended such that, rather than entitling the holder to acquire common shares of DelMar (BC), such options will entitle the holders to acquire shares of the Company. Of these options, 120,000 were cancelled during the year ended December 31, 2013.
During the year ended December 31, 2013 the Company granted an additional 2,340,000 stock options.
The following table sets forth outstanding equity awards to our named executive officers as of December 31, 2013.
| |
Option awards
|
|
Name
|
Number of
securities
underlying
unexercised
options
(#)
Exercisable
|
Number of
securities
underlying
unexercised
options
(#)
unexercisable
|
Equity incentive
plan awards:
number of
securities
underlying
unexercised
unearned
options
(#)
|
|
Option
exercise
price
(US$)
|
|
Option
expiration
date
|
|
Jeffrey Bacha(1)
|
150,000
|
-
|
-
|
|
|
0.47
|
|
2/1/2022
|
| |
132,222
|
217,778
|
-
|
|
|
1.05
|
|
8/15/2023
|
|
Dennis Brown (1)
|
150,000
|
-
|
-
|
|
|
0.47
|
|
2/1/2022
|
| |
132,222
|
217,778
|
-
|
|
|
1.05
|
|
8/15/2023
|
|
Scott Praill (1)
|
31,944
|
18,056
|
-
|
|
|
0.47
|
|
2/1/2022
|
| |
44,074
|
305,926
|
-
|
|
|
1.05
|
|
8/15/2023
|
|
(1)
|
Actual exercise price is Cdn $0.50. Price disclosed is U.S. dollar equivalent as of December 31, 2012. Options were granted on February 1, 2012 and expire on February 1, 2022.
|
The following table sets forth director compensation for the year ended December 31, 2013 (excluding compensation to the Company’s executive officers set forth in the summary compensation table above) paid by the Company (excluding any compensation included in the summary compensation table above).
| |
|
Fees
|
|
|
|
|
|
|
|
|
|
|
|
Nonqualified
|
|
|
|
|
|
|
|
| |
|
Earned
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
or
|
|
|
|
|
|
|
|
|
Non-Equity
|
|
|
Deferred
|
|
|
|
|
|
|
|
| |
|
Paid in
|
|
|
Stock
|
|
|
Option
|
|
|
Incentive Plan
|
|
|
Compensation
|
|
|
All Other
|
|
|
|
|
| |
|
Cash
|
|
|
Awards
|
|
|
Awards
|
|
|
Compensation
|
|
|
Earnings
|
|
|
Compensation
|
|
|
Total
|
|
|
Name
|
|
($)
|
|
|
($)
|
|
|
($) (1)
|
|
|
($)
|
|
|
($)
|
|
|
($)
|
|
|
($)
|
|
|
William Garner
|
|
|
58,280
|
|
|
|
-
|
|
|
|
68,520
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
126,800
|
|
|
John K. Bell
|
|
|
33,000
|
|
|
|
-
|
|
|
|
68,520
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
101,520
|
|
|
Robert J. Toth, Jr.
|
|
|
11,333
|
|
|
|
-
|
|
|
|
68,520
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
79,853
|
|
|
(1)
|
Represents the grant date fair value of 120,000 options with an exercise price of $1.05 issued on August 15, 2013. The options vested over a 12 month period and expire 10 years from the date of grant. Please see Note 8 to the financial statements.
|
The Company does not believe risks arising from its compensation policies and practices for its employees are reasonably likely to have a material adverse effect on the Company.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information, as of June 6, 2014, with respect to the beneficial ownership of the outstanding common stock by (i) any holder of more than five (5%) percent; (ii) each of the Company’s executive officers and directors; and (iii) the Company’s directors and executive officers as a group. Except as otherwise indicated, each of the stockholders listed below has sole voting and investment power over the shares beneficially owned.
|
Name of Beneficial Owner (1)
|
|
Common Stock
Beneficially Owned
|
|
|
Percentage of
Common Stock (2)
|
|
|
Directors and Officers:
|
|
|
|
|
|
|
|
Jeffrey Bacha
|
|
|
6,829,083
|
(3)
|
|
|
19.4
|
%
|
|
Dennis Brown
|
|
|
3,952,542
|
(4)
|
|
|
13.2
|
%
|
|
William Garner
|
|
|
280,000
|
(5)
|
|
|
*
|
|
|
John K. Bell
|
|
|
284,000
|
(6)
|
|
|
*
|
|
|
Scott Praill
|
|
|
510,000
|
(8)
|
|
|
1.7
|
%
|
|
Robert J. Toth, Jr.
|
|
|
163,500
|
(12)
|
|
|
*
|
|
|
All officers and directors as a group
|
|
|
|
|
|
|
34.3
|
%
|
|
Beneficial owners of more than 5%:
|
|
|
|
|
|
|
|
|
|
Valent Technologies LLC
|
|
|
2,150,000
|
(9)
|
|
|
7.3
|
%
|
|
Howard K. Fuguet (10)
|
|
|
2,500,000
|
|
|
|
8.6
|
%
|
|
Donald G. Bahout (11)
|
|
|
2,085,000
|
(7)
|
|
|
7.0
|
%
|
| |
|
|
|
|
|
|
|
|
| |
(1)
|
Except as otherwise indicated, the address of each beneficial owner is c/o DelMar Pharmaceuticals, Inc., Suite 720 - 999 West Broadway, Vancouver, British Columbia, Canada V5Z 1K5.
|
| |
(2)
|
Applicable percentage ownership is based on 28,947,760 shares of common stock outstanding as of June 6, 2014, together with securities exercisable or convertible into shares of common stock within 60 days of June 6, 2014 for each stockholder. Beneficial ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. Shares of common stock that are currently exercisable or exercisable within 60 days of June 6, 2014 are deemed to be beneficially owned by the person holding such securities for the purpose of computing the percentage of ownership of such person, but are not treated as outstanding for the purpose of computing the percentage ownership of any other person.
|
| |
(3)
|
Includes 6,287,083 shares issuable upon exchange of Exchangeable Shares (including 3,448,541 shares held in trust), and 500,000 shares issuable upon exercise of options.
|
| |
(4)
|
Includes 1,650,000 shares held by Valent, 500,000 shares issuable upon exercise of warrants held by Valent, and 500,000 shares issuable upon exercise of options.
|
| |
(5)
|
Includes 270,000 shares issuable upon exercise of options. Does not include 2,438,541 shares issuable upon exchange of Exchangeable Shares held for Mr. Garner in trust by Mr. Bacha.
|
| |
(6)
|
Includes 100,000 shares issuable upon exchange of Exchangeable Shares held by Onbelay Capital, Inc., 10,000 shares owned by Onbelay Capital, Inc., and 120,000 shares issuable upon exercise of options.
|
|
(7)
|
Includes 1,042,500 shares issuable upon exercise of warrants.
|
|
(8)
|
Includes 100,000 shares issuable upon exchange of Exchangeable Shares and 400,000 shares issuable upon exercise of options.
|
|
(9)
|
Includes 500,000 shares issuable upon exercise of warrants. Valent is owned by Dennis Brown, the Company’s Chief Scientific Officer.
|
|
(10)
|
The address of the shareholder is Ropes & Gray LLP, 800 Boylston Street, Boston MA, 02199-3600.
|
|
(11)
|
The address of the shareholder is 1059 Grand Heron Crt. West, Mobile, AL 36693.
|
|
(12)
|
Includes 120,000 shares issuable upon exercise of options.
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Certain Relationships and Related Transactions
On September 12, 2010, DelMar (BC) entered into a Patent Assignment Agreement (the “Assignment”) with Valent Technologies LLC pursuant to which Valent assigned to DelMar (BC) its rights to patent applications and the prototype drug product related to VAL-083. In accordance with the Assignment the consideration paid by DelMar (BC) was $250,000 to acquire the prototype drug product. In accordance with the terms of the Assignment, Valent is entitled to receive a future royalty (in the single digits) on certain revenues derived from the development and commercialization of VAL-083. In the event that DelMar (BC) terminates the agreement, DelMar (BC) may be entitled to receive royalties from Valent’s subsequent development of VAL-083 depending on the development milestones DelMar (BC) has achieved prior to the termination of the Assignment. The Assignment has a term (on a country-by-country basis), of the later of ten years or until patent rights covered by the Assignment no longer exist, subject to earlier termination in the event DelMar (BC) breaches its payment obligations and fails to remedy such breach within 60 days, or if either party materially beaches any of its obligations and does not cure such breach within 30 days after receipt of notice thereof.
On January 25, 2013, the Company issued to Valent 1,150,000 shares of common stock, in exchange for Valent agreeing to reduce certain royalties payable to it under the Assignment.
Pursuant to a loan agreement dated February 3, 2011, between DelMar (BC) and Valent, Valent loaned DelMar $250,000 for the purchase of the prototype drug product under the Assignment. The loan is unsecured, bears interest at 3% per year, and is payable on demand. At December 31, 2013 the loan balance, including accrued interest, was $272,372.
In addition, under the terms of the Assignment, DelMar issued to Valent warrants to acquire 500,000 common shares at an exercise price of Cdn $0.50 per upon the completion of the financing transaction that closed in February 2012.
On April 30, 2012, DelMar (BC) issued 500,000 common shares in partial settlement of accounts payable in the amount of Cdn $250,000 (U.S. $253,050) owed to Valent.
Valent, which is owned by Dr. Dennis Brown, our Chief Scientific Officer, leases facilities in California and we have access to such facilities pursuant to an informal unwritten arrangement with Valent.
For additional information see note 9 to the financial statements for the year ended December 31, 2013 included as Exhibit A to the Offer to Amend and Exercise.
William J. Garner, John K. Bell and Robert J. Toth, Jr. are independent as that term is defined under the Nasdaq Marketplace Rules.
DESCRIPTION OF SECURITIES
The Company’s authorized capital stock consists of 200,000,000 shares of common stock, par value of $0.001 per share, and 5,000,000 shares of preferred stock, par value $0.001 per share, of which 1 share has been designated Special Voting Preferred Stock. As of June 6, 2014, there are 28,947,760 shares of the Company’s common stock and 1 share of Special Voting Preferred Stock issued and outstanding.
Holders of the Company’s common stock are entitled to one vote for each share on all matters submitted to a stockholder vote. Holders of common stock do not have cumulative voting rights. Therefore, holders of a majority of the shares of common stock voting for the election of directors can elect all of the directors. Holders of the Company’s common stock representing a majority of the voting power of the Company’s capital stock issued, outstanding and entitled to vote, represented in person or by proxy, are necessary to constitute a quorum at any meeting of stockholders. A vote by the holders of a majority of the Company’s outstanding shares is required to effectuate certain fundamental corporate changes such as liquidation, merger or an amendment to the Company’s certificate of incorporation.
Holders of the Company’s common stock are entitled to share in all dividends that the board of directors, in its discretion, declares from legally available funds. In the event of a liquidation, dissolution or winding up, each outstanding share entitles its holder to participate pro rata in all assets that remain after payment of liabilities and after providing for each class of stock, if any, having preference over the common stock. The Company’s common stock has no pre-emptive rights, no conversion rights and there are no redemption provisions applicable to the Company’s common stock.
The Company’s articles of incorporation authorize the issuance of 5,000,000 shares of “blank check” preferred stock, par value $0.001 per share, in one or more series, subject to any limitations prescribed by law, without further vote or action by the stockholders. Each such series of preferred stock shall have such number of shares, designations, preferences, voting powers, qualifications, and special or relative rights or privileges as shall be determined by our board of directors, which may include, among others, dividend rights, voting rights, liquidation preferences.
Pursuant to the Certificate of Designation of the Company’s Special Voting Preferred Stock, one share of the Company’s blank check preferred stock has been designated as Special Voting Preferred Stock. The Special Voting Preferred Stock votes as a single class with the common stock and is entitled to a number of votes equal to the number of Exchangeable Shares of Exchangeco outstanding as of the applicable record date (i) that are not owned by the Company or any affiliated companies and (ii) as to which the holder has received voting instructions from the holders of such Exchangeable Shares in accordance with the Trust Agreement.
The Special Voting Preferred Stock is not entitled to receive any dividends or to receive any assets of the Company upon any liquidation, and is not convertible into common stock of the Company.
The voting rights of the Special Voting Preferred Stock will terminate pursuant to and in accordance with the Trust Agreement. The Special Voting Preferred Stock will be automatically cancelled at such time as the share of Special Voting Preferred Stock has no votes attached to it.
Common stock issued upon exchange of Exchangeable Shares and upon the exercise of Exchange Agreement Warrants held by Canadian residents may be subject to statutory hold periods in accordance with applicable Canadian securities laws.
For a description of our warrants, please see the Offer to Amend and Exercise.
Indemnification of Directors and Officers
Neither our Articles of Incorporation nor Bylaws prevent us from indemnifying our officers, directors and agents to the extent permitted under the Nevada Revised Statute ("NRS"). NRS Section 78.7502 provides that a corporation shall indemnify any director, officer, employee or agent of a corporation against expenses, including attorneys' fees, actually and reasonably incurred by him in connection with any the defense to the extent that a director, officer, employee or agent of a corporation has been successful on the merits or otherwise in defense of any action, suit or proceeding referred to Section 78.7502(1) or 78.7502(2), or in defense of any claim, issue or matter therein.
NRS 78.7502(1) provides that a corporation may indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, except an action by or in the right of the corporation, by reason of the fact that he is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses, including attorneys' fees, judgments, fines and amounts paid in settlement actually and reasonably incurred by him in connection with the action, suit or proceeding if he: (a) is not liable pursuant to NRS 78.138; or (b) acted in good faith and in a manner which he reasonably believed to be in or not opposed to the best interests of the corporation, and, with respect to any criminal action or proceeding, had no reasonable cause to believe his conduct was unlawful.
NRS Section 78.7502(2) provides that a corporation may indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the corporation to procure a judgment in its favor by reason of the fact that he is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against expenses, including amounts paid in settlement and attorneys' fees actually and reasonably incurred by him in connection with the defense or settlement of the action or suit if he: (a) is not liable pursuant to NRS 78.138; or (b) acted in good faith and in a manner which he reasonably believed to be in or not opposed to the best interests of the corporation. Indemnification may not be made for any claim, issue or matter as to which such a person has been adjudged by a court of competent jurisdiction, after exhaustion of all appeals there from, to be liable to the corporation or for amounts paid in settlement to the corporation, unless and only to the extent that the court in which the action or suit was brought or other court of competent jurisdiction determines upon application that in view of all the circumstances of the case, the person is fairly and reasonably entitled to indemnity for such expenses as the court deems proper.
NRS Section 78.747 provides that except as otherwise provided by specific statute, no director or officer of a corporation is individually liable for a debt or liability of the corporation, unless the director or officer acts as the alter ego of the corporation. The court as a matter of law must determine the question of whether a director or officer acts as the alter ego of a corporation.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling us pursuant to the foregoing provisions, we have been informed that, in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, we will, unless in the opinion of our counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by us is against public policy as expressed hereby in the Securities Act and we will be governed by the final adjudication of such issue.
RECENT SALES OF UNREGISTERED SECURITIES
DelMar (BC) issued 500,000 units on October 3, 2011, 100,000 units on October 7, 2011, and 50,000 units on November 11, 2011, at a purchase price of Cdn $0.50 per unit or total consideration of Cdn $325,000 (U.S. $310,570). Each unit consisted of one common share and share purchase warrant. Of the total consideration of Cdn $325,000, Cdn $125,000 (U.S. $118,925) relates to non-cash consideration received for the provision of services by officers and directors of DelMar (BC) and the settlement of accounts payable with an officer of DelMar (BC).
DelMar (BC) issued 4,150,000 units on January 23, 2012, 560,000 units on February 27, 2012, and 50,000 units on May 10, 2012, at a purchase price Cdn $0.50 per unit or total consideration of Cdn $2,380,000 (U.S. $2,361,353). The proceeds from the issuance of 3,000,000 of these units were held in escrow pursuant to an exclusive option investment agreement. Subsequently, DelMar (BC) elected to allow the option to expire and the related units were cancelled and the funds returned to the subscriber.
Included in the total consideration of Cdn $2,380,000 (US $2,361,353) was Cdn $180,000 (US $180,144) relating to non-cash consideration received for the provision of services by officers and directors of DelMar (BC). All of the units issued pursuant to the consulting agreements were issued in February 2012.
Between June 1, 2012 and January 1, 2013 inclusive DelMar (BC) issued 160,000 common shares for services.
On April 30, 2012, DelMar (BC) issued 500,000 common shares in settlement of accounts payable in the amount of Cdn $250,000 (U.S. $253,050) owed to Valent.
On January 25, 2013 the Company entered into and closed an exchange agreement, with DelMar BC, 0959454 B.C. Ltd., Callco, Exchangeco, and securityholders of DelMar (BC). Pursuant to the Exchange Agreement, (i) the Company issued 4,340,417 shares of common stock to the U.S. Holders in exchange for the transfer to Exchangeco of all 4,340,417 outstanding common shares of DelMar (BC) held by the U.S. Holders, (ii) the Canadian Holders received, in exchange for the transfer to Exchangeco of all 8,729,583 outstanding common shares of DelMar (BC) held by the Canadian Holders, 8,729,583 Exchangeable Shares of Exchangeco, and (iii) outstanding warrants to purchase 3,360,000 common shares of DelMar (BC) and outstanding options to purchase 1,020,000 common shares of DelMar (BC) were deemed to be amended such that, rather than entitling the holder to acquire common shares of DelMar (BC), such options and warrants will entitle the holders to acquire shares of common stock of the Company. The Canadian Holders will be entitled to require Exchangeco to redeem (or, at the option of the Company or Callco, to have the Company or Callco purchase) the Exchangeable Shares, and upon such redemption or purchase to receive an equal number of shares of common stock of the Company.
On January 25, 2013, January 31, 2013, February 8, 2013, February 21, 2013, February 28, 2013, March 1, 2013, and March 6, 2013, the Company entered into and closed a series of subscription agreements with accredited investors, pursuant to which the Company issued an aggregate of 13,125,002 Units at a purchase price of $0.80 per Unit, for aggregate gross proceeds of $10,500,000. Each Unit consists of one share of common stock and one five-year warrant to purchase one share of common stock at an exercise price of $0.80.
The Company retained Charles Vista, LLC as the Placement Agent for the Private Offering. The Company paid the Placement Agent a cash fee of $1,050,000 (equal to 10% of the gross proceeds), a non-accountable expense allowance of $315,000 (equal to 3% of the gross proceeds), and a one-year consulting fee of $60,000. Also, the Company issued to the Placement Agent five-year warrants to purchase 5,250,000 shares of common stock (equal to 20% of the shares of common stock (i) included as part of the Units sold in the Private Offering and (ii) issuable upon exercise of the Investor Warrants) at an exercise price of $0.80, exercisable on a cash or cashless basis.
On January 25, 2013, the Company, Callco, Exchangeco and Computershare Trust Company of Canada (the “Trustee”) entered into a voting and exchange trust agreement (the “Trust Agreement”). Pursuant to the Trust Agreement, Company issued one share of Special Voting Preferred Stock (the “Special Voting Share”) to the Trustee, and the parties created a trust for the Trustee to hold the Special Voting Share for the benefit of the holders of the Exchangeable Shares (other than the Company and any affiliated companies) (the “Beneficiaries”). Pursuant to the Trust Agreement, the Beneficiaries will have voting rights in the Company equivalent to what they would have had they received shares of common stock in the same amount as the Exchangeable Shares held by the Beneficiaries.
On January 25, 2013, the Company issued 1,150,000 shares of common stock to Valent, a company owned by Dennis Brown, the Company’s Chief Scientific Officer, in exchange for Valent agreeing to reduce certain royalties payable to it pursuant to a patent assignment agreement between Valent and DelMar (BC).
On April 17, 2013, the Company issued an aggregate of 555,000 shares of restricted common stock to consultants for services.
During the three months ended September 30, 2013, the Company issued an aggregate of 1,469,810 shares of common stock, including 40,000 shares of common stock for services, 206,000 shares upon the exercise of 206,000 warrants for no additional consideration, 123,810 shares upon the cashless exercise of 200,000 warrants, and 1,100,000 shares upon the exchange of Exchangeable Shares. On September 12, 2013, the Company issued five-year warrants to purchase 300,000 shares of common stock for services. The warrants are exercisable on a cashless basis at an exercise price of $1.76.
During the three months ended December 31, 2013, the Company issued 15,000 shares of common stock upon the exercise of 15,000 warrants for no additional consideration and 255,000 shares of common stock upon the exchange of 255,000 Exchangeable Shares.
During the period from January 1, 2014 to April 14, 2014 the Company issued 305,313 shares of common stock upon the exercise of warrants, including 20,000 shares for no additional consideration, 8,000 shares for proceeds of CDN $4,000, and 277,313 shares for proceeds of $221,850. In addition, during the period from January 1, 2014 to April 14, 2014 the Company issued 250,000 shares of common stock for services, and 205,000 shares of common stock upon the exchange of 205,000 Exchangeable Shares. On June 6, 2014, the Company issued 3,652,211 shares of common stock upon the exercise of warrants for net proceeds of $2,255,240.
The transactions described above were exempt from registration under Section 4(a)(2) of the Securities Act and/or under Regulation S promulgated by the SEC.
ADDITIONAL INFORMATION FURNISHED BY REFERENCE
The Company has included in its Schedule TO, the accompanying Offer to Amend and Exercise and this Supplemental Company Information the information required by Form 10. The Company incorporates by reference into this Supplemental Company Information the documents listed below and filed by the Company pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act.
| |
•
|
|
our Quarterly Report on Form 10-Q for the fiscal quarter ended March 31, 2014, filed with the SEC on May 15, 2014;
|
| |
•
|
|
our Annual Report on Form 10-K for the fiscal year ended December 31, 2013, filed with the SEC on March 10, 2014;
|
| |
•
|
|
our Current Reports on Form 8-K filed with the SEC on June 9, 2014; and
|
| |
•
|
|
the description of our common stock contained in our registration statement on Form 8-A filed with the SEC on September 9, 2012.
|
These documents, and all exhibits attached thereto, can be accessed electronically at no cost on the SEC’s website at www.sec.gov. In addition, the Company will provide each holder of an Investor Warrant a copy of any or all of these documents and any other information that has been incorporated by reference into this Supplemental Company Information upon written or oral request at no cost to the requester. Requests should be directed to: DelMar Pharmaceuticals, Inc. Suite 720 - 999 West Broadway, Vancouver, British Columbia CANADA V5Z 1K5; Attn: Corporate Secretary.
The following exhibit index shows those exhibits incorporated herein by reference:
|
Exhibit Number
|
|
Description
|
|
2.1
|
|
Exchange Agreement, dated January 25, 2013, among the Company, Exchangeco, Callco, DelMar (BC) and securityholders of DelMar (BC) (1)
|
|
3.1
|
|
Articles of Incorporation of the Company (2)
|
|
3.2
|
|
Articles of Merger of the Company (3)
|
|
3.3
|
|
Certificate of Designation of Special Voting Preferred Stock of the Company (3)
|
|
3.4
|
|
Bylaws of the Company (2)
|
|
3.5
|
|
Amendment to Bylaws of the Company (4)
|
|
10.1
|
|
Intercompany Funding Agreement, dated January 25, 2013, between the Company and Exchangeco (1)
|
|
10.2
|
|
Support Agreement, dated January 25, 2013, among the Company, Exchangeco and Callco (1)
|
|
10.3
|
|
Voting and Exchange Trust Agreement, dated January 25, 2013, among the Company, Callco, Exchangeco, and the Trustee (1)
|
|
10.4
|
|
Form of Subscription Agreement (1)
|
|
10.5
|
|
Form of Registration Rights Agreement (1)
|
|
10.6
|
|
Form of Investor Warrant (1)
|
|
10.7
|
|
Form of Dividend Warrant (1)
|
|
10.8 †
|
|
Memorandum of Understanding and Collaboration Agreement between Guangxi Wuzhou Pharmaceutical (Group) Co. Ltd. and DelMar (BC) (1)
|
|
10.9 †
|
|
Patent Assignment Agreement, dated September 12, 2010, between DelMar (BC) and Valent (5)
|
|
10.10
|
|
Amendment, dated January 21, 2013, to Patent Assignment Agreement, dated September 12, 2010, between DelMar (BC) and Valent (5)
|
|
10.11
|
|
Loan Agreement, dated February 3, 2011, between DelMar (BC) and Valent (5)
|
|
10.12
|
|
Consulting Agreement, dated August 1, 2011, between DelMar (BC) and Jeffrey Bacha (5)
|
|
10.13
|
|
Consulting Agreement, dated August 1, 2011, between DelMar (BC) and Dennis Brown (5)
|
|
10.14
|
|
Consulting Agreement, dated August 1, 2011, between DelMar (BC) and William Garner (5)
|
|
10.15
|
|
Consulting Agreement, dated February 1, 2013, between DelMar (BC) and Scott Praill (5)
|
|
16
|
|
Letter from John Kinross-Kennedy (1)
|
|
21
|
|
Subsidiaries (6)
|
† Confidential treatment has been granted for certain confidential portions of this exhibit pursuant to Rule 24b-2 under the Exchange Act. In accordance with Rule 24b-2, these confidential portions have been omitted from this exhibit and filed separately with the Commission
(1) Filed as exhibit to 8-K filed on January 31, 2013 and incorporated herein by reference.
(2) Filed as an exhibit to S-1 filed August 17, 2010 and incorporated herein by reference.
(3) Filed as an exhibit to 8-K filed January 23, 2013 and incorporated herein by reference.
(4) Filed as an exhibit to 8-K filed February 14, 2013 and incorporated herein by reference.
(5) Filed as exhibit to 8-K/A filed on March 14, 2103 and incorporated herein by reference.